DC Bead™ (including DC Bead™ M1)
Microsphere
DC Bead™ is a loadable microsphere product primarily intended as an embolic agent for the local treatment of malignant hypervascularised tumour(s) in the liver and also intended to embolise the vessels supplying malignant colorectal cancer metastasised to the liver (mCRC).
Key Resources
Explore
Product Description
DC Bead is a preformed, deformable microsphere consisting of a biocompatible, sulphonate-modified, N-Fil hydrogel.
DC Bead Features
- Calibrated size to ensure predictable embolization1
- Compressible design for smooth catheter delivery1
- Compatible with a large range of microcatheters1
- Blue tinted beads for enhanced visualization1
- Spherical shape for optimal control and delivery1
DC Bead M1
70-150μm beads allow:
- Controlled distal embolization results in a more homogeneous distribution in the targeted tissue2
- Greater likelihood of tumor necrosis2
- Less opportunity for the development of collateral circulation2
Drug-Eluting Bead: DEBDOX™
DC Bead™ are compatible with doxorubicin for the local treatment of tumours in patients with hepatocellular carcinoma (HCC). Doxorubicin can be loaded prior to embolisation and as a secondary action, will elute a local, controlled and sustained dose to the tumour after embolisation.
Drug-Eluting Bead: DEBIRI™
DC Bead™ can be loaded prior to embolisation and then as a secondary action, elute a local, controlled and sustained dose to the liver metastases from colorectal cancer after embolisation.
- Biocompatibles data on file
- Mueller PR and Adams A (editors). Interventional Oncology: A Practical Guide for the Interventional Radiologist, Chapter 7, Principles of Embolization. Springer Science + Business Media, LLC 2012.2
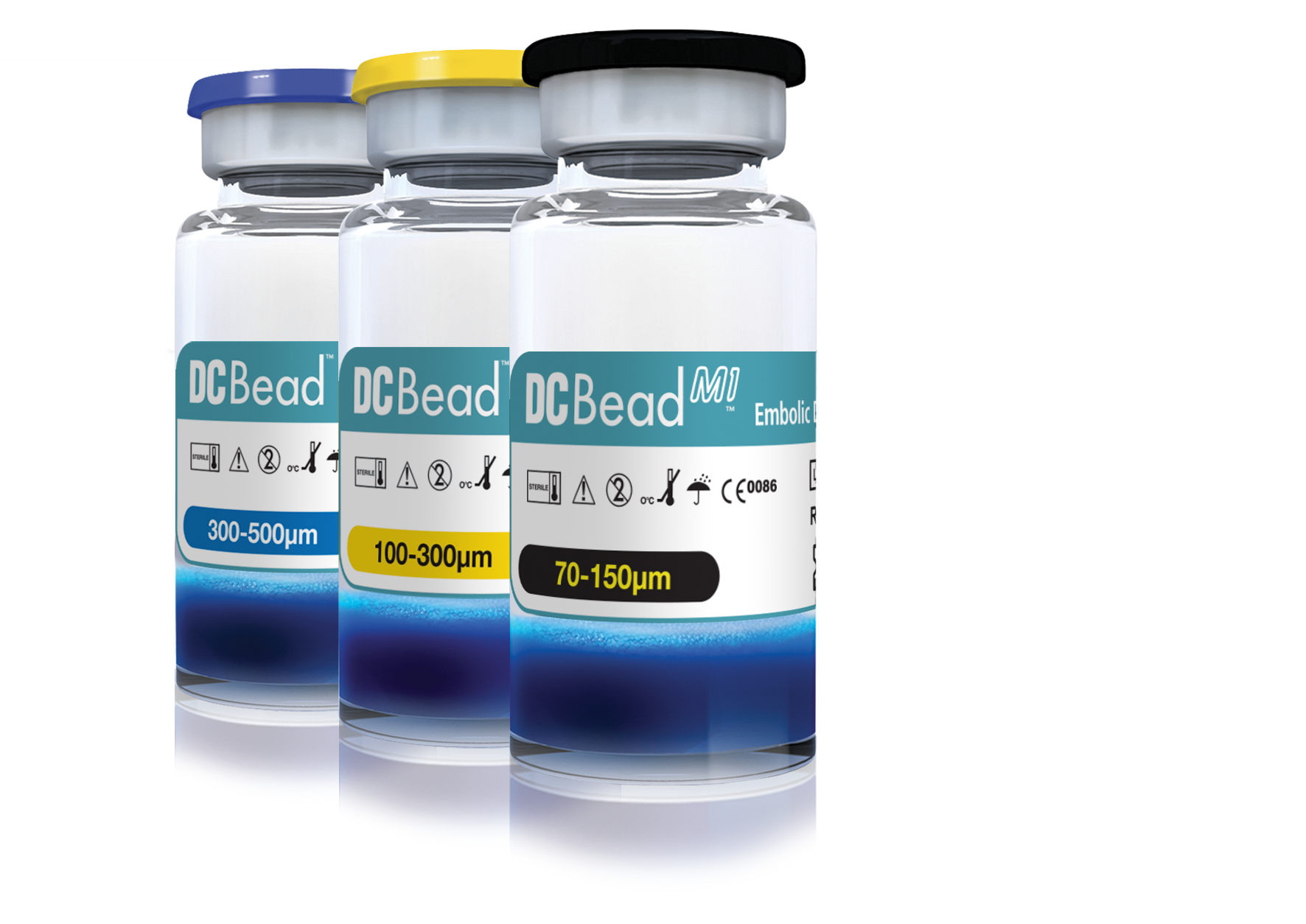
Product Specifications and Sizes
- DC Bead M1 is available in one size: 70-150μm
- DC Bead is available in 3 sizes from 100μm to 500μm
DC BEAD M1 70-150MM | DC BEAD 100-300MM | DC BEAD 300-500MM | |
| Approximate number of beads in 2ml vial* | 1,300,000 | 200,000 | 38,000 |
| Size | Label Color |
| 70-150µm | Black |
| 100-300µm | Yellow |
| 300-500µm | Blue |
* Theoretical calculation based on the average diameters of a normal distribution of beads in a vial.
1. Biocompatibles data on file.
CAUTION: The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings and instructions for use can be found in the product labelling supplied with each device. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France. 2020 Copyright © Boston Scientific Corporation or its affiliates. All rights reserved. 93-204