The ULTIMA trial is the first (and only) head-to-head prospective, randomized-controlled trial that showed EKOS™ is more effective than anticoagulation alone and just as safe. This controlled trial of ultrasound-assisted catheter-directed thrombolysis for acute intermediate-risk Pulmonary Embolism was done in 2014.
Nils Kucher, MD et al. Circulation 2014; 129: 479-486
Clinical significance
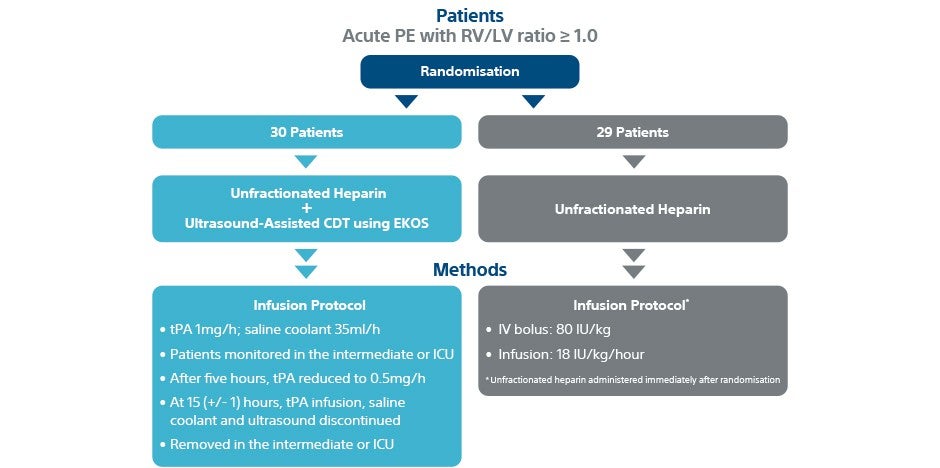
Trial overview
- Prospective, multi-center, randomised, controlled trial
- 59 patients with acute intermediate-risk PE
- 8 centres in Germany and Switzerland
- Infusion time: 15 hours. Total dose: 20 mg
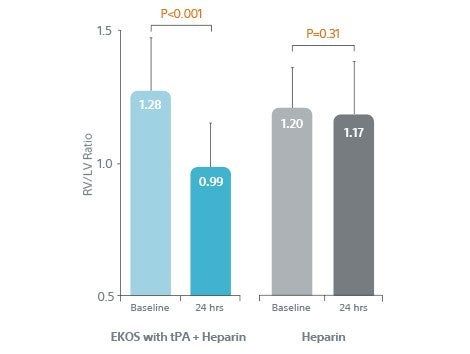
- 23% reduction (p<0.001) in RV/LV ratio from baseline vs. 2.5% (p=.031) in Heparin group
- No major bleeds, deaths or recurrent VTE at 90 days
- 0 ICH
Patients
Key results
- RV/LV ratio significantly improved at 24 hours.
- Reduction in RV/LV ratio significantly greater at 24 hours and improved at 90 days.
- No deaths or significant bleeding complications.
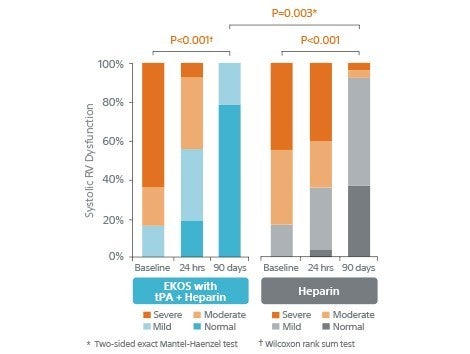
- Systolic RV dysfunction significantly improved.
RV/LV Ratio Significantly
Improved at 24 Hours
Reduction in RV/LV Ratio
Significantly
Greater at 24 Hours and Improved at 90 days

Systolic RV Dysfunction
Significantly improved
No Deaths or Significant Bleeding Complications
| Clinical outcomes at 90 days | EKOS with tPA + Heparin N = 30 | Heparin N = 29 | P-value | ||
| Death | 0 | 0% | 1* | 0% | 0.49 |
| Recurrent venous thromboembolism | 0 | 0% | 0 | 0% | 1 |
| Major bleeding | 0 | 0% | 0 | 0% | 1 |
| Major bleeding | 3** | 10% | 1 | 3% | 0.61 |
*Rehospitalisation and death from advanced pancreatic cancer
**Two patients with transient mild hemoptysis without medical intervention, one patient with groin hematoma requiring manual compression
✞One patient with transient anal bleeding following endoscopic removal of colon polyp
Trial conclusions
The ULTIMA trial showed that a fixed-dose EKOS regimen was superior to anticoagulation alone in improving right ventricular dysfunction at 24 hours without an increase in bleeding complications.
Trial bibliography
2014 - ULTIMA: Randomised, Controlled Trial of Ultrasound-Assisted Catheter-Directed Thrombolysis for Acute Intermediate-Risk Pulmonary Embolism.
Nils Kucher, MD et al. Circulation 2014; 129: 479-486
[https://pubmed.ncbi.nlm.nih.gov/24226805]
Caution:
The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.
















