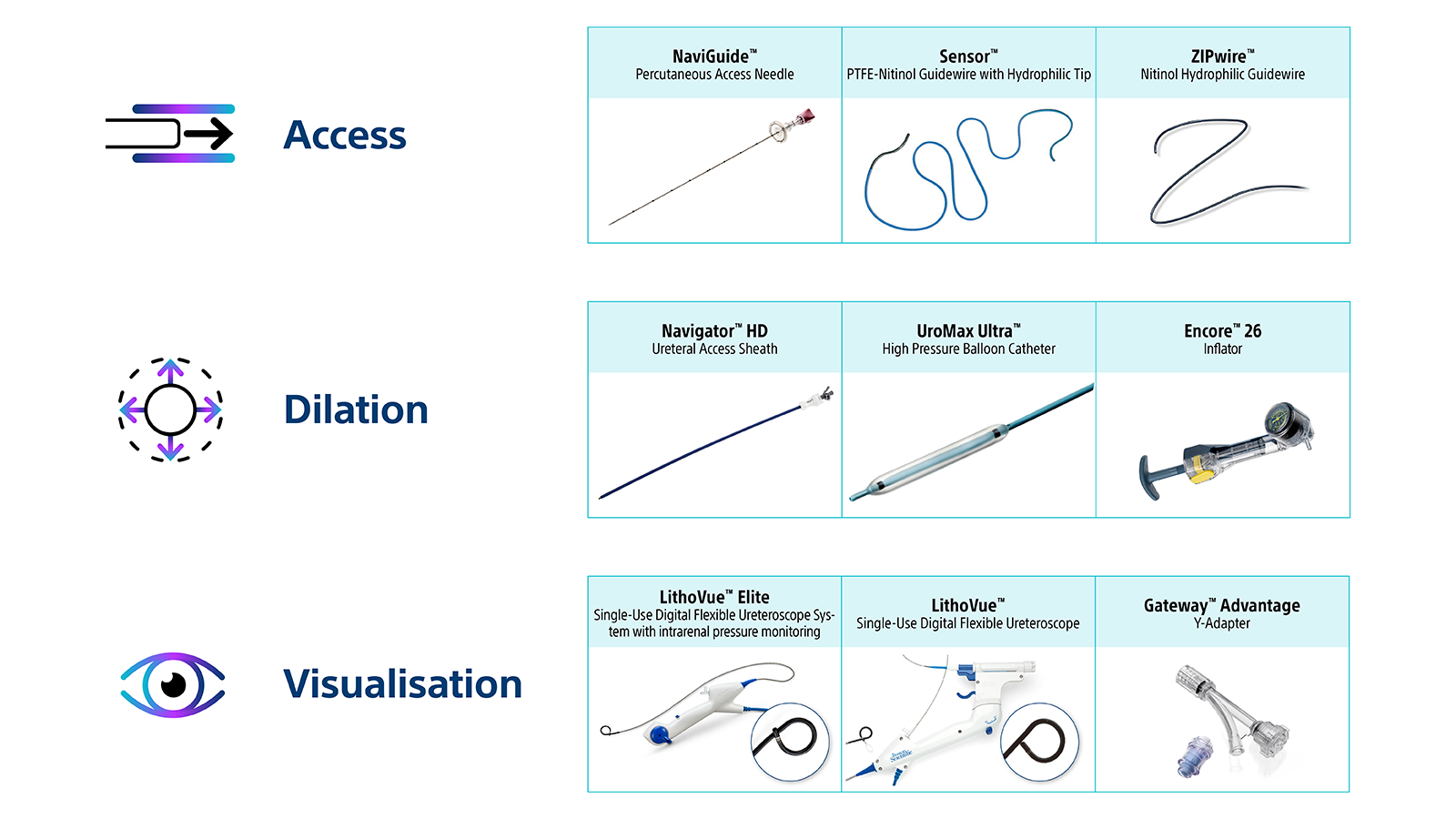
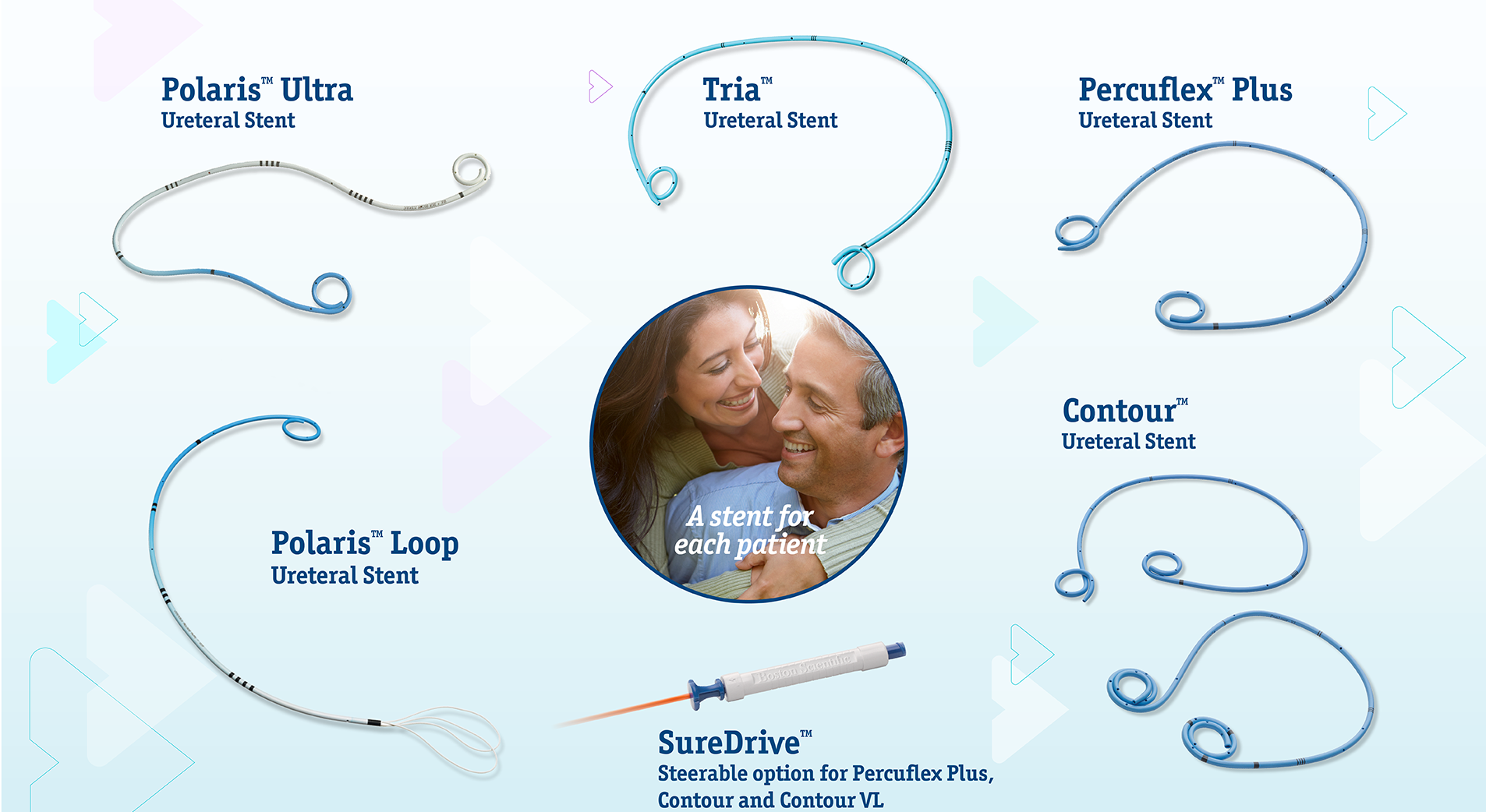
Boston Scientific offers you a versatile solution that may allow success regardless of technique preference.1-11, 17, 18, 23 We are also offering a wide range of products built for each patient, despite their distinct anatomical needs8-12,14-20,23, and a versatile portfolio for kidney stone management no matter the size, type, or location.1, 8-14, 20-23
- Designed to simplify tasks and increase efficiency.1-11, 17, 18,23
- Designed to meet patients’ needs and preferences.8-12, 14-20,23
- For various stone sizes, locations and compositions.1, 8-14, 17, 18, 20-23