Expand access to curative surgery options

Many HCC patients are faced with minimal options at diagnosis. TheraSphereTM Y90 Therapy can help your patients gain access to curative treatment options, no matter what the BCLC stage.
Target lesion response early-on
Patients with very early- to early-stage HCC not favourable for ablation
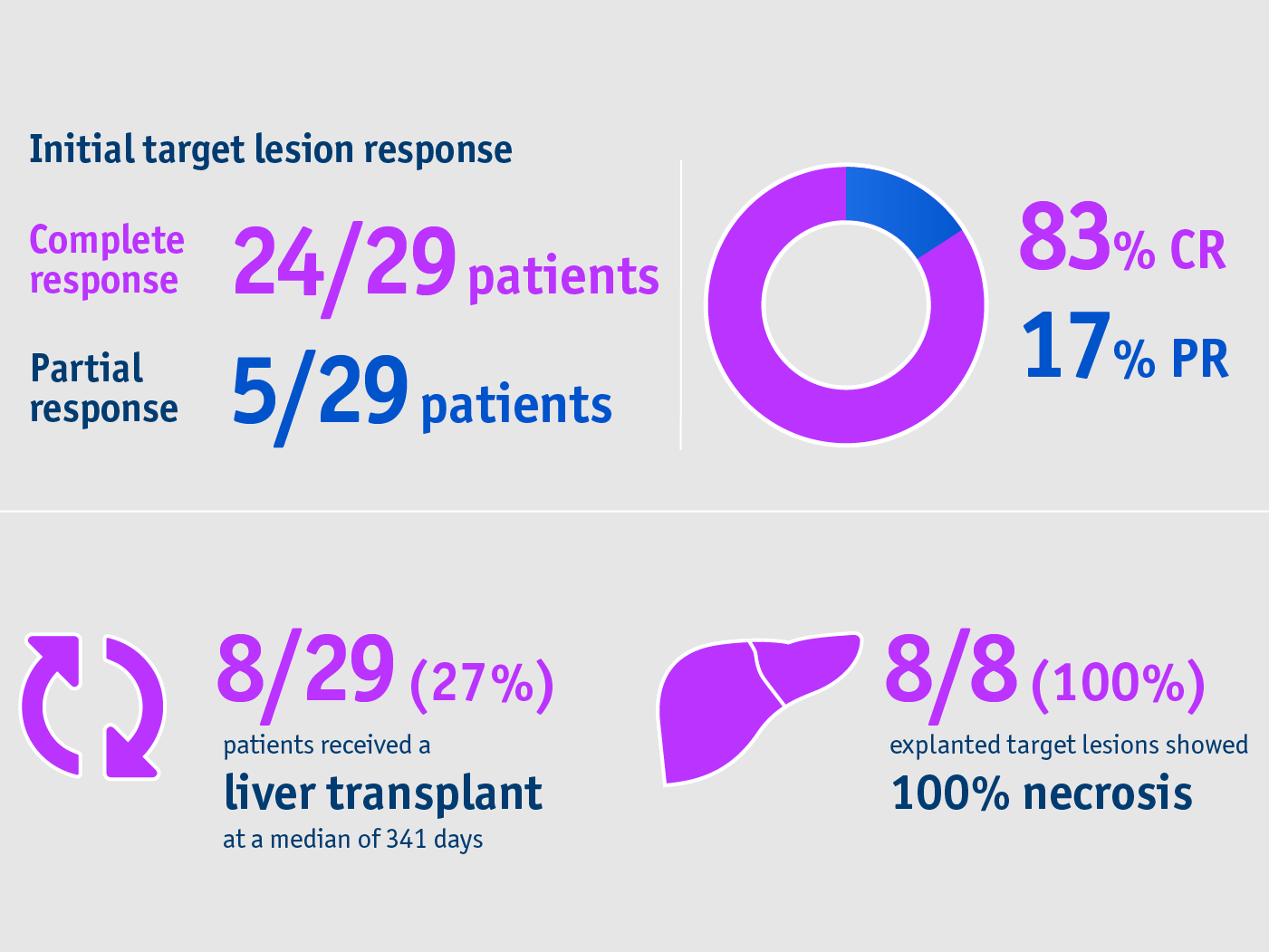
➣ 83% CR at the target lesion site following TheraSphereTM Y90 Therapy1
➣ 27% of patients received a liver transplant following TheraSphereTM Y90 Therapy1
Early-stage HCC in the RASER trial1
- Complete response rates
- Summary
- Study design
Day 0: Patient with T3 HCC prior to treatment
Day 42: Patient received orthotopic liver transplant
Explant specimen showed complete necrosis of the target tumour
Follow-up: Successful transplant and patient showed no evidence of recurrence
RASER trial (Kim et al. 2022)
• Prospective
• Open-label
• Single-centre (USA)
• Single-arm
Patients with very early- to early-stage solitary HCC with unfavourable location for ablation
• Tumour diameter ≤3 cm
• Child-Pugh A–B7
• ECOG score 0
Prolong progression free survival
Patients with intermediate-stage HCC not amenable to curative treatment
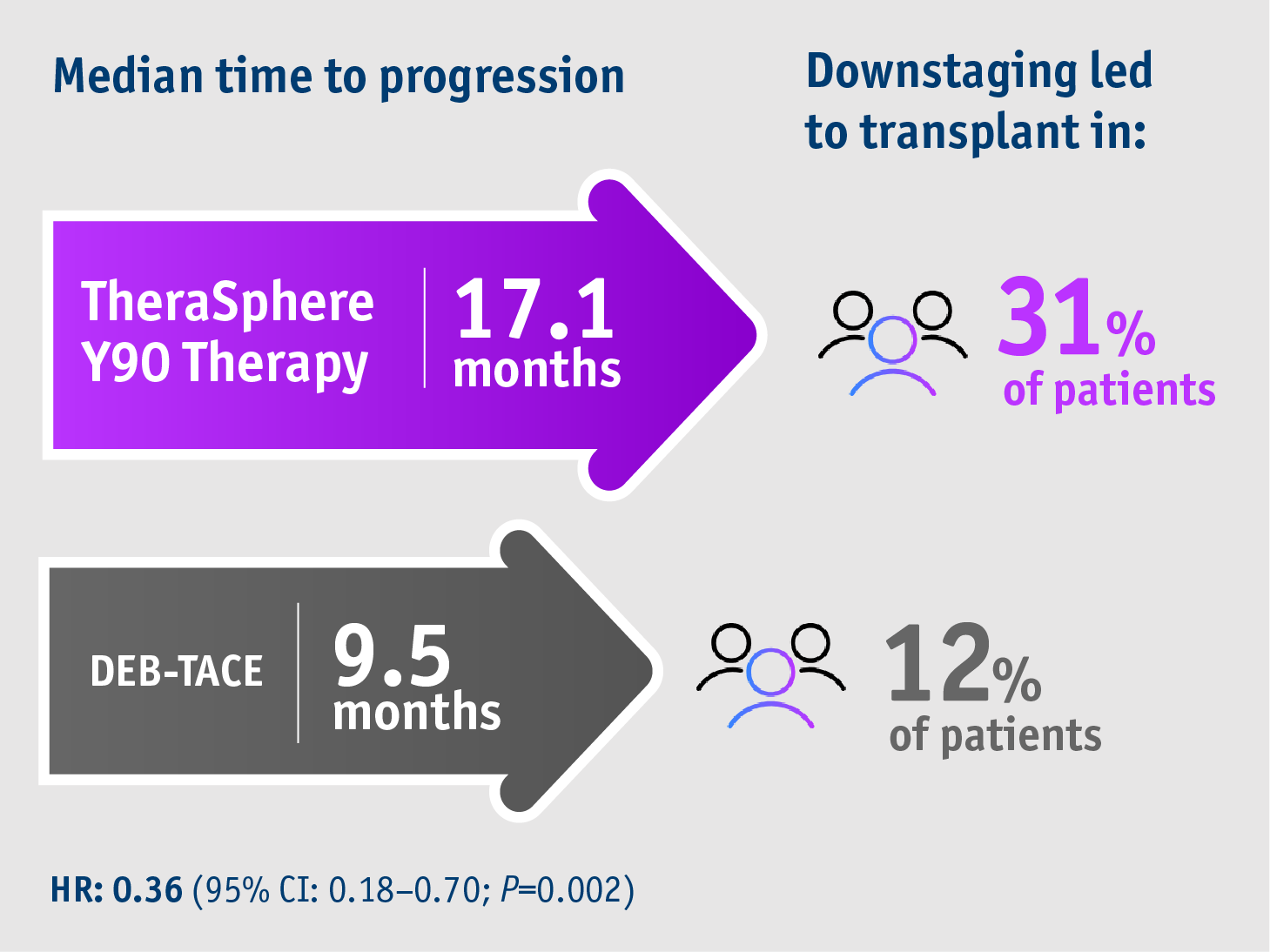
➣ 17.1 months median TTP allowed >2x patients to access liver transplant following TheraSphereTM Y90 Therapy vs DEB-TACE3
Phase II TRACE trial3
- Time to progression
- Liver transplant
- Summary
- Study design
Time to overall tumour progression in the ITT group
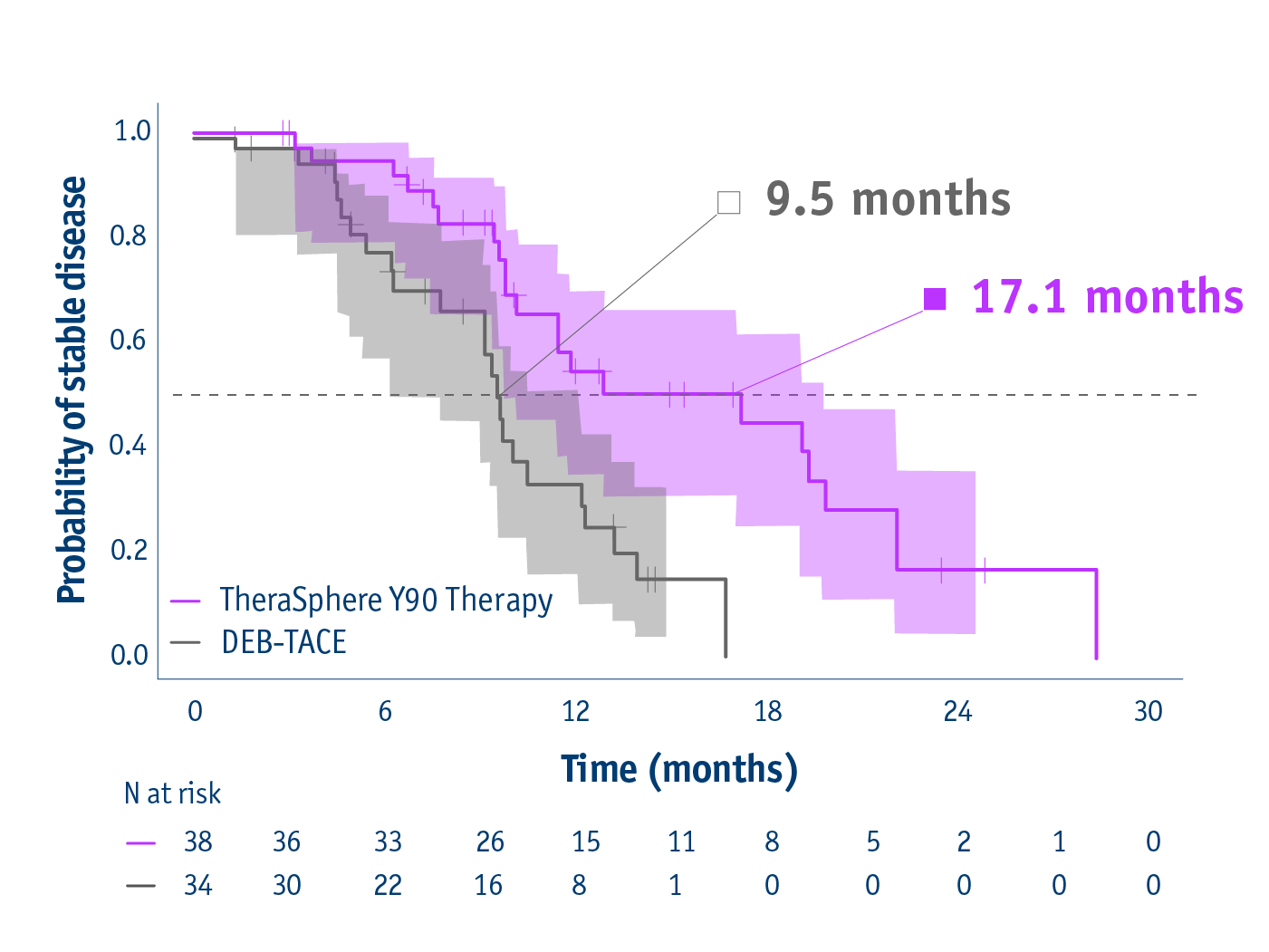
- 17.1 months median TTP with TheraSphereTM Y90 Therapy
(95% CI: 8.9–25.4) - 9.5 months median TTP with DEB-TACE
(95% CI: 8.8–10.2) - HR: 0.36
(95% CI: 0.18–0.70; P=0.002)
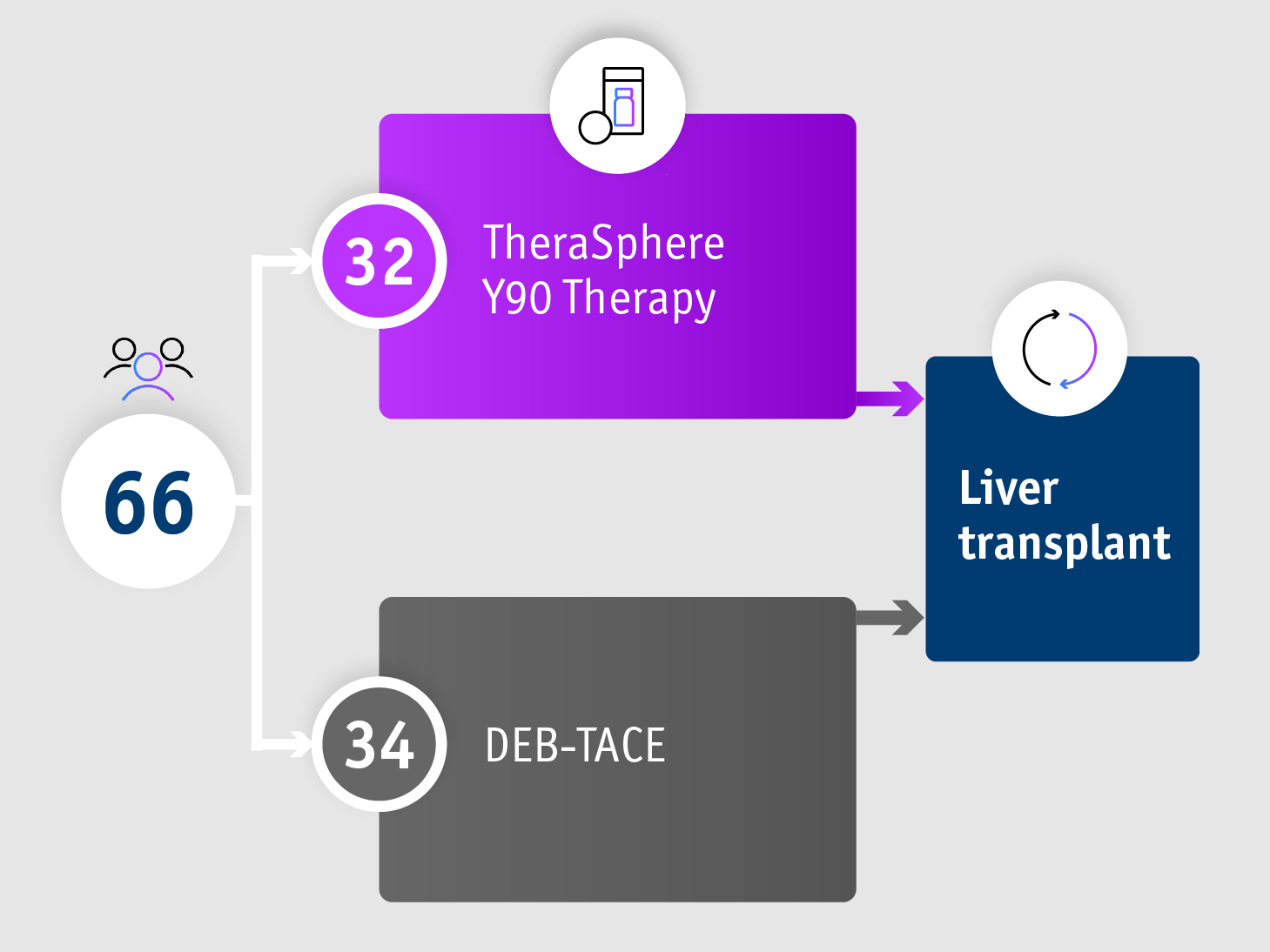
TRACE trial (Dhondt et al. 2022)
• Phase II trial
• Open-label
• Single-centre (Belgium)
• Randomised
• Controlled
Patients with HCC not amenable to curative treatment
• BCLC A/B
• ECOG PS 1
• Segmental PVT
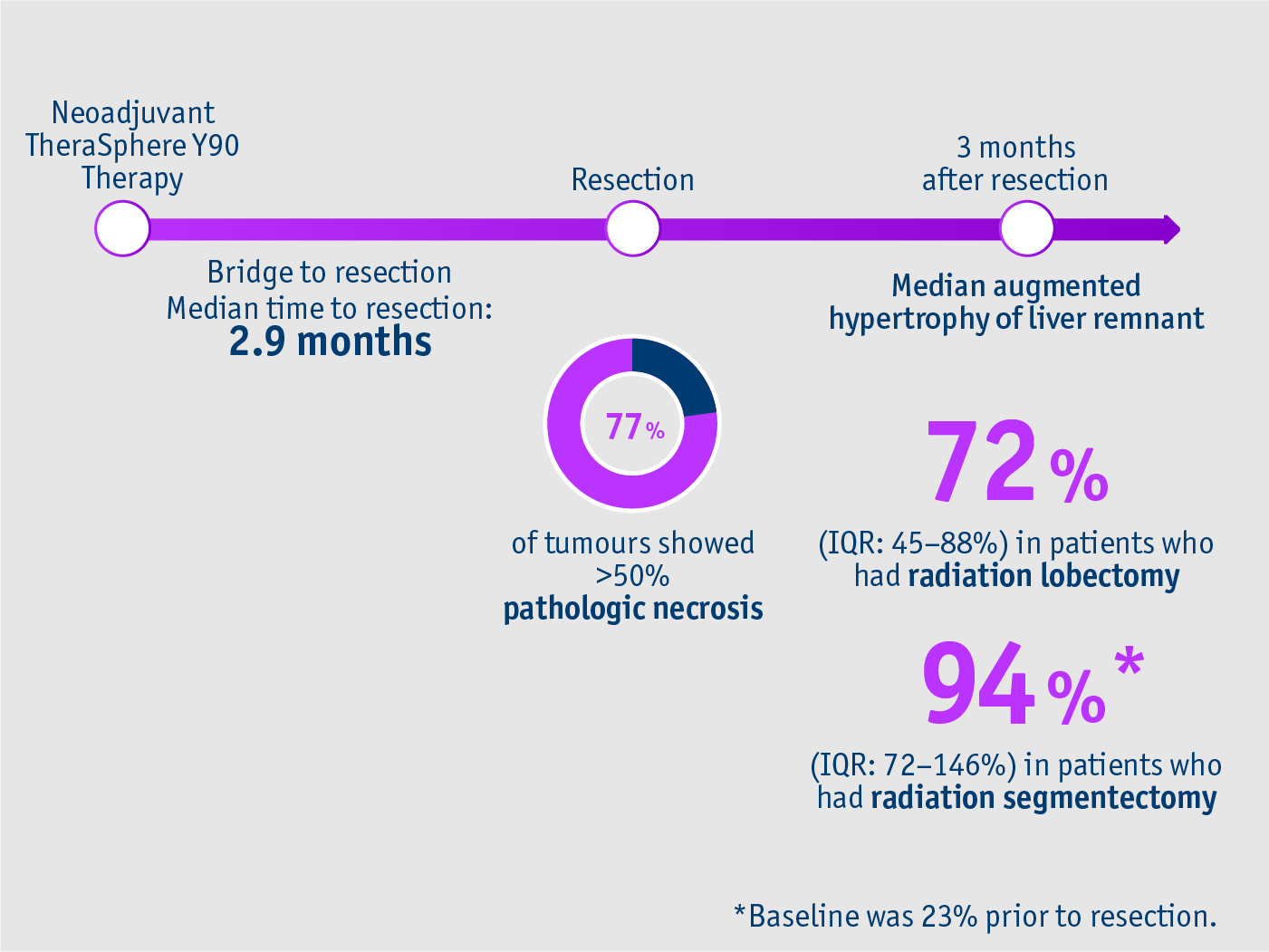
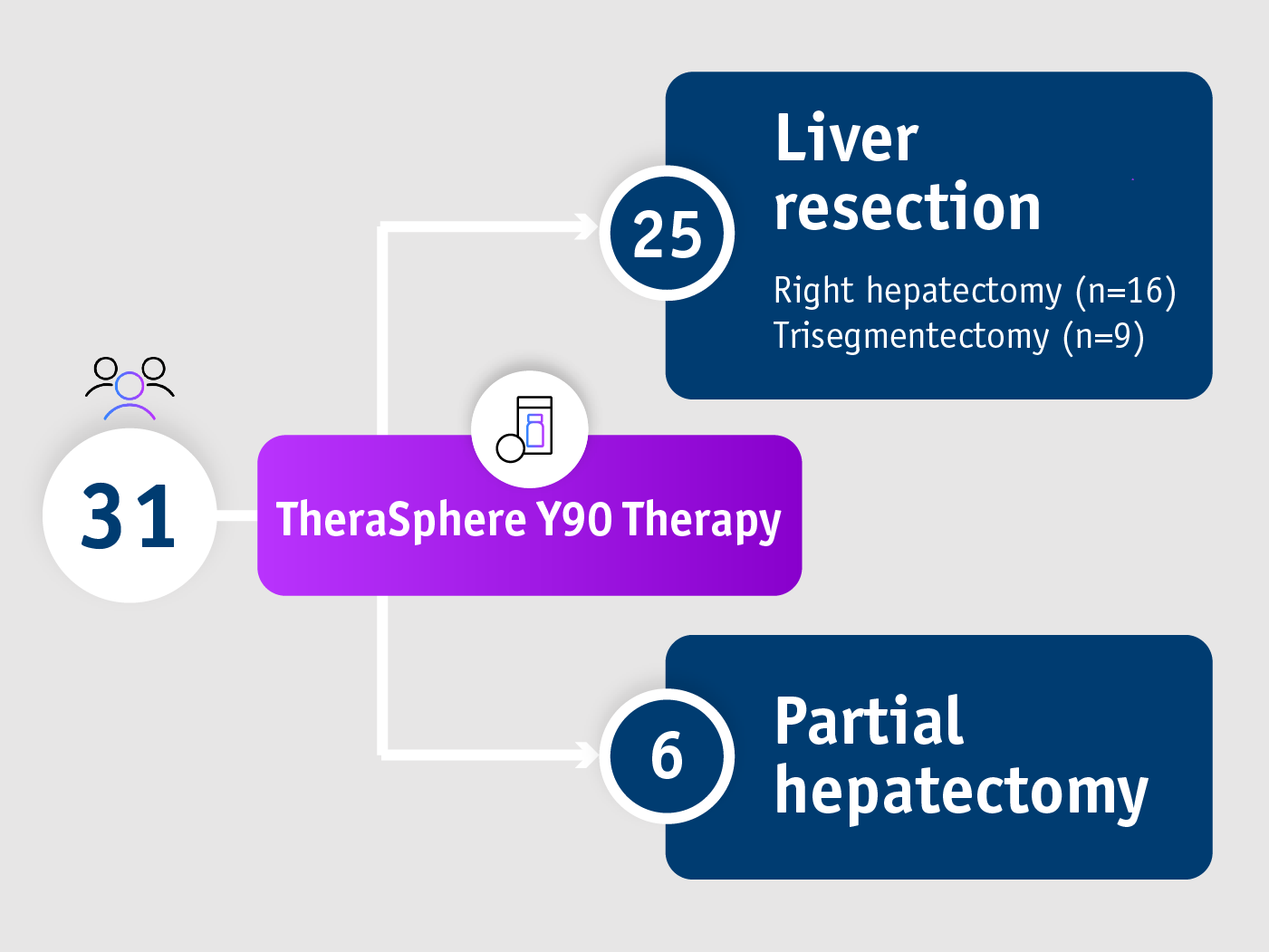
Bridge safely and promote future liver remnant
For patients with BCLC A, B and C HCC (CP ≤7)
➣ 77% of tumours showed more than 50% pathologic necrosis at resection following TheraSphereTM Y90 Therapy4
Longitudinally clinical and radiological outcomes study Gabr et al. 20184
- Future liver remnant
- Clinical outcomes
- Summary
- Study design
Did you know?
According to BCLC Guidelines, SIRT is included into clinical decision making for very early and early stage HCC5,6

Gabr et al. 2018
• Retrospective
• Single centre (USA)
Patients with unilobar HCC
• Preserved liver function
• Child-Pugh ≤ B7
• No portal vein thrombus or metastatic disease
Gain access to curative surgery
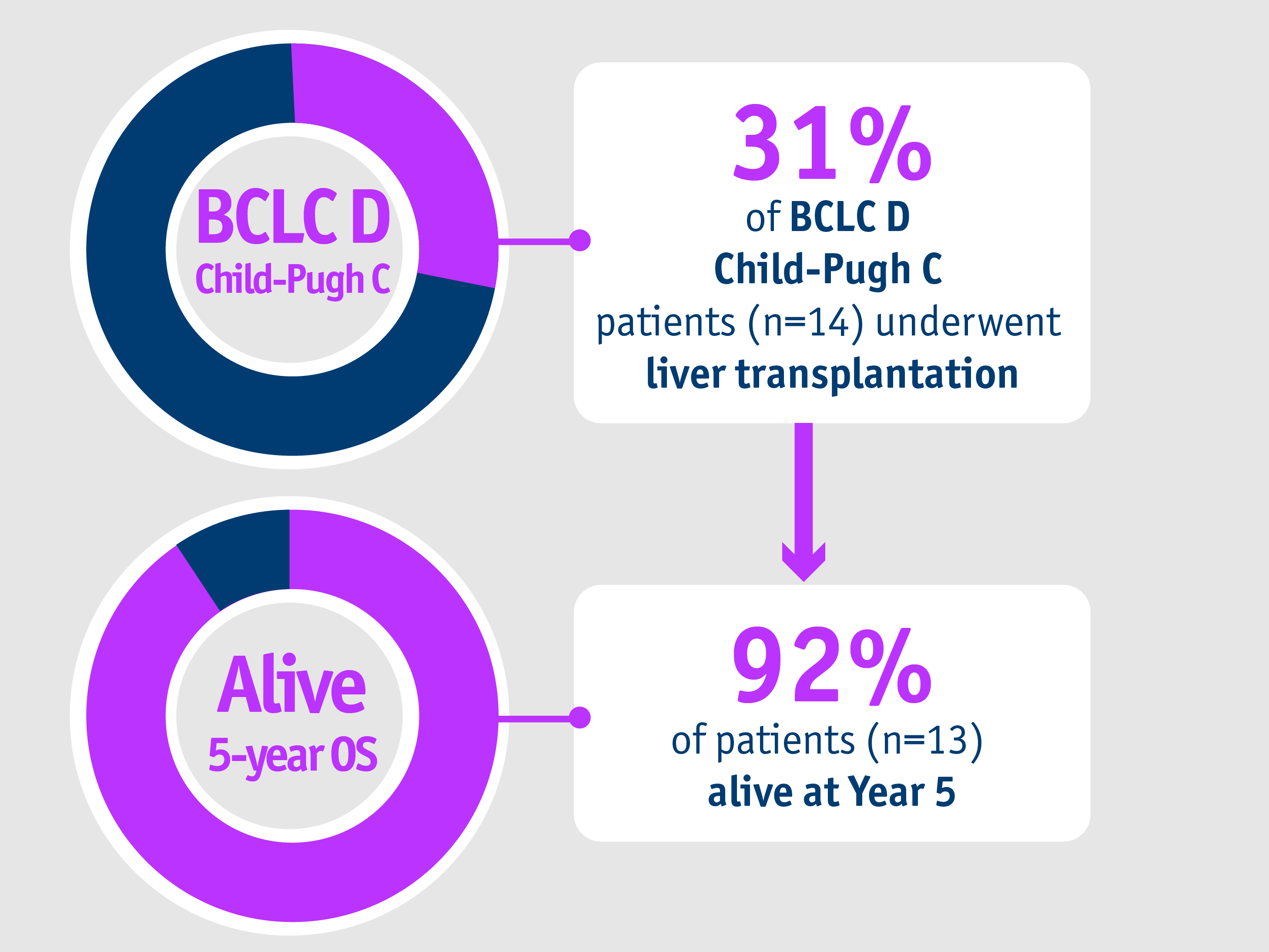
Patients with advanced stage HCC
➣ ~1/3 of patients with BCLC D, CP-C stage HCC accessed liver transplantation following TheraSphereTM Y90 Therapy7
Large single-centre HCC cohort: Salem et al. 20187
- Palliative to curative
- Summary
- Study design
Palliative care has traditionally been recommended for BCLC D patients
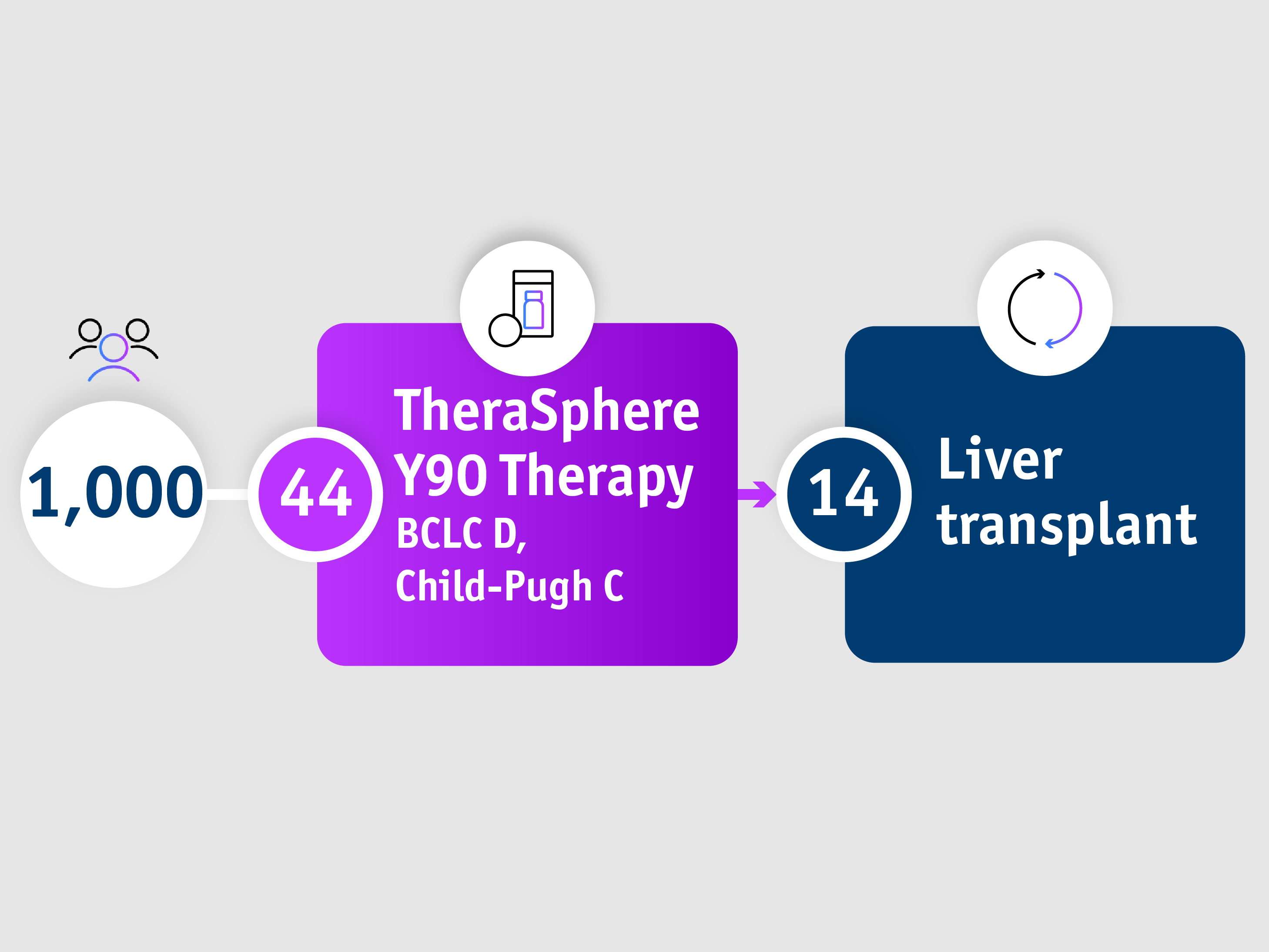
Salem et al. 2018
• From 2003 to 2017
• 1,000 HCC patients were treated with Y-90 glass microspheres as part of a prospective cohort study (the largest single-centre cohort conducted) with 1,577 total treatments (median: 1, range: 1-8)
Baseline characteristics:
• ECOG: 0 (56%); 1 (40%); 2 (4%)
• Child-Pugh: A (51%); B (45%); C (4%)
• BCLC: A (26%); B (15%); C (54%); D (4%)
References
1.Kim E, et al. Radiation segmentectomy for curative intent of unresectable very early to early stage hepatocellular carcinoma (RASER): a single-centre, single-arm study. Lancet Gastroenterol Hepatol. 2022 Sep;7(9):843-850.
2.Kulik LM, et al. Use of yttrium-90 microspheres (TheraSphere) in a patient with unresectable hepatocellular carcinoma leading to liver transplantation: a case report. Liver Transpl. 2005 Sep;11(9):1127-1131.
3.Dhondt E, et al. 90Y Radioembolization versus Drug-eluting Bead Chemoembolization for Unresectable Hepatocellular Carcinoma: Results from the TRACE Phase II Randomized Controlled Trial. Radiology. 2022 Jun;303(3):699-710.
4.Gabr A, et al. Outcomes of Surgical Resection after Radioembolization for Hepatocellular Carcinoma. J Vasc Interv Radiol. 2018 Nov;29(11):1502-1510.e1.
5.Reig M, et al. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J Hepatol. 2022 Mar;76(3):681-693.
6.Vogel A, Martinelli E; ESMO Guidelines Committee. Electronic address: clinicalguidelines@esmo.org; ESMO Guidelines Committee. Updated treatment recommendations for hepatocellular carcinoma (HCC) from the ESMO Clinical Practice Guidelines. Ann Oncol. 2021 Jun;32(6):801-805.
7.Salem R, et al. Institutional decision to adopt Y90 as primary treatment for hepatocellular carcinoma informed by a 1,000-patient 15-year experience. Hepatology. 2018;68(4):1429-1440.
Abbreviations
AE, adverse event; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; EASL, European Association for the Study of the Liver; ECOG, Eastern Cooperative Oncology Group; HCC, hepatocellular carcinoma; HR, hazard ratio; ITT, intention to treat; mRECIST, modified Response Evaluation Criteria in Solid Tumors; ORR, objective response rate; OS, overall survival; PVT, portal vein thrombosis; RFS, recurrence-free survival; TACE, transarterial chemoembolisation; TTP, time to progression; UNOS, United Network for Organ Sharing; Y90, yttrium-90.
Caution:
The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.
















