Are HCC treatment strategies doing enough to preserve liver function?3,4
Many HCC treatment options are unpredictable and have adverse effects on liver function and healthy liver tissue. This risk is compounded in patients with a cirrhotic background.3,4

Uncover HCC treatment strategies to preserve liver function3,5,6

TheraSphereTM Y90 Therapy
as standalone curative or neoadjuvant therapy in HCC.
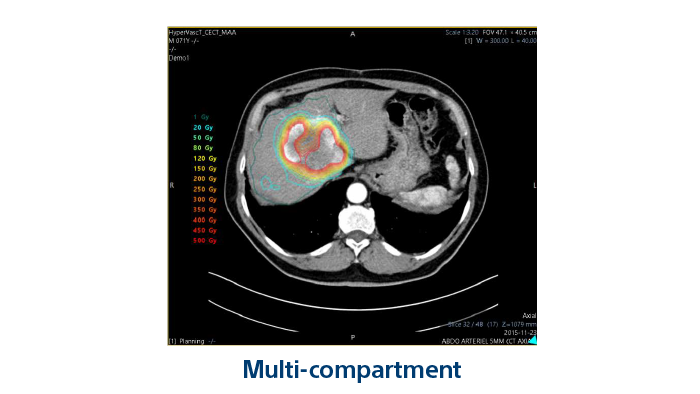
Personalised and versatile TheraSphereTM Y90 Therapy empowers multidisciplinary teams to control liver cancer while preserving liver function across BCLC stages A to D3,5,15
References
1.Gabr A, al. Liver Transplantation Following Yttrium‐90 Radioembolization: 15‐year Experience in 207‐Patient Cohort. Hepatology 2021;73(3):998-1010.
2.DOSISPHERE-01: Garin E, et al. Personalised versus standard dosimetry approach of selective internal radiation therapy in patients with locally advanced hepatocellular carcinoma (DOSISPHERE-01): a randomised, multicentre, open-label phase 2 trial. Lancet Gastroenterol Hepatol. 2021 Jan;6(1):17-29.
3.European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69(1):182-236.
4.Cheng CJ, et al. Radiation-induced liver disease after radiotherapy for hepatocellular carcinoma: clinical manifestation and dosimetric description. Radiother Oncol. 2002;63(1):41-45.
5.Riaz A, Gates VL, Atassi B et al. Radiation segmentectomy: A novel approach to increase safety and efficacy of radioembolization. Int J Radiat Oncol Biol Phys 2011;79(1):163–71.
6.Lewandowski RJ, et al. A comparative analysis of transarterial downstaging for hepatocellular carcinoma: chemoembolization versus radioembolization. Am J Transplant. 2009;9(8):1920-1928.
7.Quirk M, et al. Management of hepatocellular carcinoma with portal vein thrombosis. World J Gastroenterol. 2015; 21(12): 3462-3471.
8.Boston Scientific. Most studied Y-90 HCC therapy. Available at: https://www.bostonscientific.com/en-US/medical-specialties/interventional-radiology/interventional-oncology/therasphere/clinical-data.html. Accessed October 2023.
9.Schwander B, Klesper K, Rossol S et al. Poster presentation EE566 at ISPOR EU 2022. Available at: https://www.ispor.org/heor-resources/presentations-database/presentation/euro2022-3568/118940. Accessed October 2023.
10.Rognoni C, et al. Cost-effectiveness analysis of personalised versus standard dosimetry for selective internal radiation therapy with TheraSphere in patients with hepatocellular carcinoma. Front Oncol. 2022 Aug 29;12:920073.
11.Manas D, Bell JK, Mealing S et al. The cost-effectiveness of TheraSphere in patients with hepatocellular carcinoma who are eligible for transarterial embolization. Eur J Surg Oncol 2020.
12.Chaplin S, Taylor M, Lapon J, White J. Economic evaluation of glass yttrium-90 microspheres versus sorafenib for the treatment of advanced hepatocellular carcinoma: cost effectiveness analysis in the United Kingdom. Cardiovasc Intervent Radiol 2015;38:S279-80
13.LEGACY: Salem R, et al. Yttrium-90 Radioembolization for the Treatment of Solitary, Unresectable HCC: The LEGACY Study. Hepatology. 2021; 74(5): 2342–2352.
14.Mazzaferro V, et al. Yttrium-90 Radioembolization for Intermediate-Advanced Hepatocellular Carcinoma: A Phase 2 Study. Hepatology. 2013;57(5):1826-1837.
15.Kim HC. Radioembolization for the treatment of hepatocellular carcinoma. Clin Mol Hepatol. 2017;23(2):109-114.
Abbreviations
BCLC, Barcelona Clinic Liver Cancer; HCC, hepatocellular carcinoma; mOS, median overall survival; Y90, yttrium-90.
Caution:
The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.
TheraSphere is a registered trademark of Theragenics Corporation, used under license by Boston Scientific Medical Device Limited, a wholly owned indirect subsidiary of Boston Scientific Corporation.
CЄ 0123
















