Rotational Atherectomy Systems
ROTAPRO™ and ROTABLATOR™ Rotational Atherectomy Systems
Key Resources
Explore
Clinical Article: Contemporary Rotational Atherectomy
Rotational Atherectomy: you will never regret using it!
EuroIntervention, Dec 2016
Dr. Teresa Strisciuglio (Naples. Italy) and Dr. Emanuele Barbato (Aalst, Belgium) illustrate the advantages deriving from the contemporary rotational atherectomy technique.
Rotational Atherectomy has recently gone through a surge of interest. It has evolved from “plaque debulking” to what nowadays is called “plaque modification”. In fact, the contemporary rotational atherectomy technique aims to smoothen the lumen and disconnect the calcified coronary ring, thus leaving the way clear for further balloon dilatation and stent implantation
Today rotablation techniques use smaller burrs and smaller burr-to-artery ratio, lower rotational speed and pecking motion tecnique.

University of Naples Federico II, Naples, Italy
These are currently considered best practices in using rotablation leading to a decrease in complications and improving outcomes.
Current practice tends to use rotational atherectomy as a bailout only after inadequate dilation of the vessel. The bail-out attitude tends to be replaced by a more judicious elective indication to rotational atherectomy in parallel with operator experience and with increasing confidence with the technique. In fact, you will never regret using rotational atherectomy, but you often regret not having used it!
| Contemporary Rotational Atherectomy Technique | |
| Arterial access | Radial (6-7.5 Fr) or femoral (6-8 Fr), depending´upon burr size requirement and operator experience |
| Guiding catheter | Single curve with strong support. Operator preference but stable catheter position required |
| Guidewire | Rotawire placement not always straightforward. Use of regular wire placement, with exchange using microcatheter placement often required |
| Burr size | Plaque modification with small burrs (1.25 mm to 1.5 mm) as initial strategy is default position. A step-up approach is encouraged to limit debrissize and complications |
| Ablation speed | Plaque modification usually achieved at low speeds (135,000 to 180,000 rpm) to reduce risk of complications |
| Temporary pacemaker | Smaller burrs at lower speeds have led to lower incidence of transient heart block. Many operators use atropine to treat, avoiding any complications of temporary pacemaker placement |
| Rotablation flush | Rotablation cocktail with verapamil, nitrates and heparin in saline recommended |
ROTATE Multicentre Registry
In-hospital and midterm clinical outcomes of rotational atherectomy followed by stent implantation: the ROTATE multicentre registry
EuroIntervention, Dec 2016
Rotational atherectomy (RA) results in high procedural success rates with acceptable short- and longer-term major adverse cardiac event (MACE) rates considering the severity of patient and lesion characteristics. Multivariable analysis identified dialysis to be an independent predictor for both in-hospital and follow-up MACE. Drug-eluting stent (DES) use was associated with a reduction in follow-up MACE. RA use may also be useful to facilitate the treatment of complex coronary lesions with newer technology (e.g., bioresorbable scaffolds). Larger, randomised studies are required to investigate the efficacy of RA further, for both existing and new indications.
In-Hospital and Midterm Outcomes of Rotational Atherectomy (RA)

In-Hospital and Midterm Outcomes of Rotational Atherectomy (ROTATE Multi-Center Registry)
| Patient/Lesion Characteristics | N=985 (1176 lesions) |
| Diabetes Mellitus | 34.5% |
| ACS | 26.2% |
| Three-vessel disease | 37.2% |
| Haemodialysis | 7.6% |
| Bifurcation lesions | 25.2% |
| Target Vessel LAD | 48.2% |
| Outcomes | N=985 |
| In-hospital MACE* | 8.3% |
| 1-year follow-up MACE* | 16.0% |
| 2-year follow-up MACE** | 24.9% |
| 2-year ST (definite/probable) | 1.8% |
Advantages of Planned Versus Provisional Rotational Atherectomy (RA) for Severely Calcified Coronary Lesions
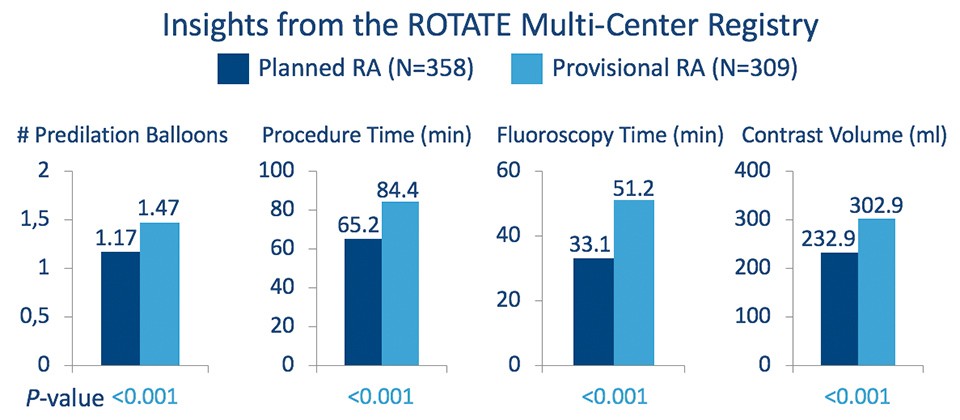
Planned RA was associated with a reduction in resources compared to Provisional RA in the ROTATE Multi-Center Registry
The primary endpoint of in-hospital MACE* tended to be better in the planned RA group compared to the provisional RA group (unadjusted OR: 0.76; 95% CI: 0.44-1.31, P=0.32, and adjusted OR: 0.59, 95% CI: 0.33-1.05, P=0.07). *MACE defined as the composite endpoint of all-cause death, follow-up myocardial infarction, and target lesion revascularization; Kawamoto, H et. al. Catheter Cardiovasc Interv 2016;88(6):881-889.















