S-ICD System
Subcutaneous Implantable Defibrillator
2-year Results from a POOLED Analysis of the IDE Study and EFFORTLESS Registry (Published in JACC in early 2015)1
• 882 patients, Average Follow-Up: 651 days
• Largest patient cohort, most comprehensive data and longest follow-up period further demonstrates the worldwide safety and efficacy of the S-ICD System in a large diverse population
• Combining the studies provides a unique opportunity to evaluate:
- Complications
- Spontaneous events
US IDE Study (Published in CirculaIon 2013)2
• 321 patients, Average Follow-Up: 11 months
• Completed in 2011 and was the cornerstone for US FDA approval
• Primary Safety Endpoint: 180-Day S-ICD System Complication Free Rate compared to prespecified goal of 79 %
• Primary Efficacy Endpoint: Induced VF conversions of 4 attempts compared with prespecified goal of 88 %
EFFORTLESS Registry Interim Results (Published in European Heart Journal in early 2014)3
• 456 patients, Average Follow-Up: 558 days
• Ongoing Registry In Europe and New Zealand
• Primary Outcome Measures:
- Perioperative S-ICD System Complication Free Rate
- 360 Day S-ICD System Complication Free Rate
• Inappropriate shocks for AF / SVT
The S-ICD System has been implanted in a BROAD RANGE OF PATIENTS
| Demographic | N (%) |
|---|---|
| Age (years) | 50.3 ± 16.9* |
| Male | 636 (72.5 %) |
| Ischemic | 330 (37.8 %) |
| Genetic | 58 (6.7 %) |
| Idiopathic VF | 40 (4.6 %) |
| Channelopathies | 90 (10.3 %) |
| NYHA Classification II-IV | 327 (37.5 %) |
| Arial Fibrillation | 143 (16.4 %) |
| Previous Defibrillator | 120 (13.7 %) |
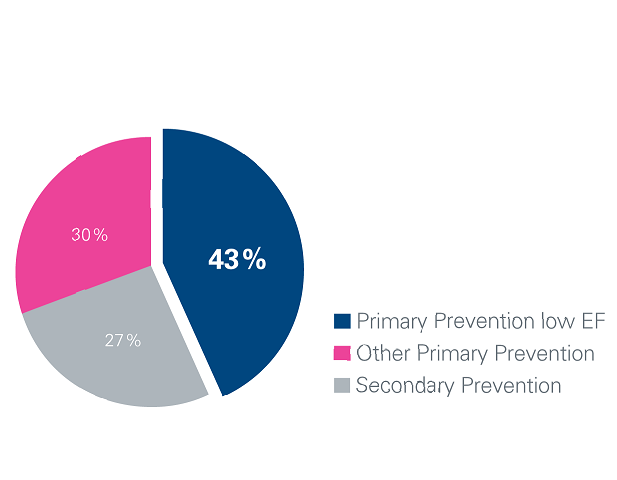
43 % of the study population were primary prevention patients with an EF ≤ 35 % 1
Data has demonstrated a SAFE solution for sudden cardiac death
Complications
ZERO1
Electrode Failures, Systemic Blood Infections, Endocarditis or Cardiac Injuries
The lack of problematic consequences of endovascular complications including systemic infections could be a factor in the observed low mortality rate (Tarakji KG et al Europace 2014).
In the IDE Study, there were no explants due to infection in the last 2 / 3 implantations.
Data has demonstrated an EFFECTIVE solution for sudden cardiac arrest
Conversion Efficacy of Induced Arrhythmias
98.6%1
The S-ICD System consistently demonstrates effective conversions of induced arrhythmias.
Conversion Efficacy of Discrete Spontaneous Arrhythmias
98.2 %1
Clinical conversion of spontaneous arrhythmias was achieved in all patients.
First Shock Efficacy
91.1 %1
The S-ICD System first shock
conversion efficacy is in line with
rates published for TV-ICDs.3