Express™ LD
Iliac and Biliary Premounted Stent System
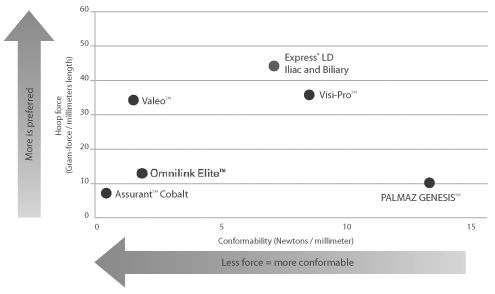
Only Express LD Iliac and Biliary Stent was engineered to provide the perfect balance of compression resistance and conformability—without trade-off.
Key Resources
Explore
Product Details
Express LD Stent System features a patented Tandem Architecture™ Stent Design
Micro™ Elements engineered to provide:
- Flexibility during placement
- Conformability upon deployment
- Maximum apposition
Macro™ Elements engineered for:
- Balanced compression resistance
- Strength—even in arterial bifurcations

Clinical Information
MELODIE Trial Summary
MELODIE is a prospective, multi-center, single arm study to obtain additional data on the safety and efficacy of the Express™ Vascular LD stent implantation in the treatment of stenosed or occlusive atherosclerotic disease (de novo or restenotic) in iliac arteries (common or external).
MELODIE Trial Objective
The objective was to demonstrate non-inferiority of the Express™ LD Stent for the treatment of atherosclerotic iliac artery lesions as compared to an objective performance criterion* (OPC), with a primary endpoint of mean percent luminal diameter loss at 6 months.
*Note: the OPC=15%, δ=5%.
Primary Endpoint
Definition
- Angiographic mean percent loss of luminal diameter at 6 months post-procedure, defined as [POST MLD1 - FUP MLD2 / POST MLD] x 100
1Post MLD = Post-procedure minimum lumen diameter.
2FUP MLD = Follow-up minimum lumen diameter at 6 months.
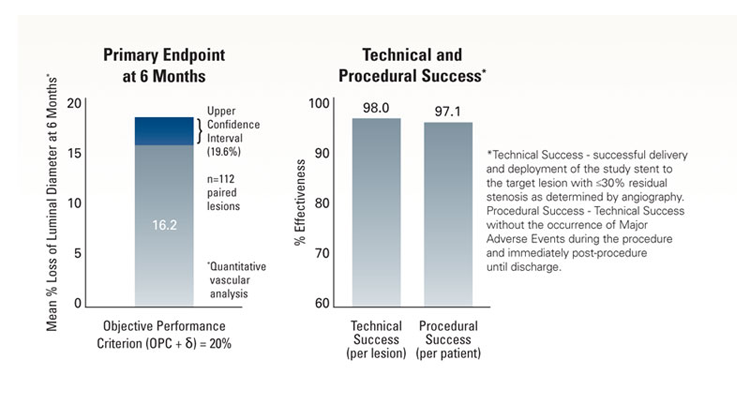
Outcome
- Mean percent luminal diameter loss at 6 months = 16.2% ± 18.4%
- Upper Confidence Int = 19.6%
- Clinical follow-up rate at 6 months = 92.6% (138/150)
- This rate was non-inferior to the Objective Performance Criterion = δof 20%
24-Month Results
- The Express LD Stent = 8.9%, 10.2% TLR at 1 and 2 years and no device/procedure-related death
- Sustained Stent Target Lesion Patency measured by CTA with 97.2% at 1 year and 94.1% at 2 years
Tools & Resources
-

-
 Aug 24, 2017
Aug 24, 2017MRI Compatibility for PI Products
Magnetic Resonance Imaging (MRI) Safety for Boston Scientific Peripheral Products PDF, 272.0 KB













