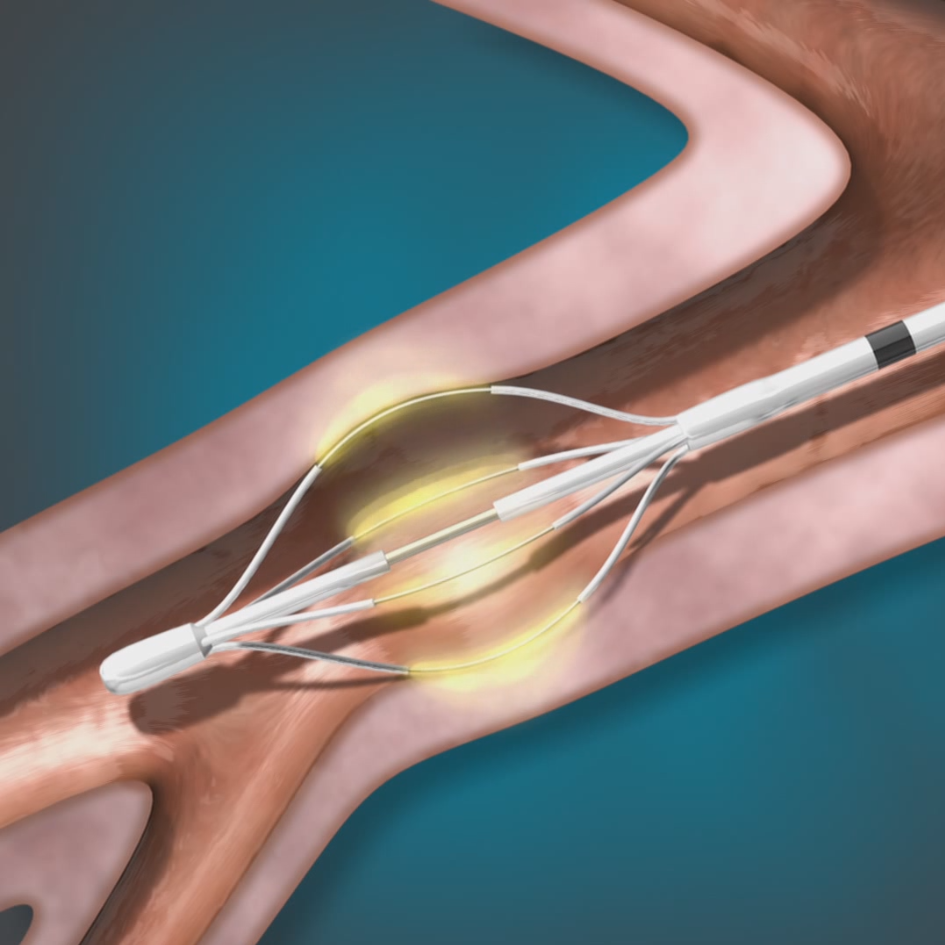
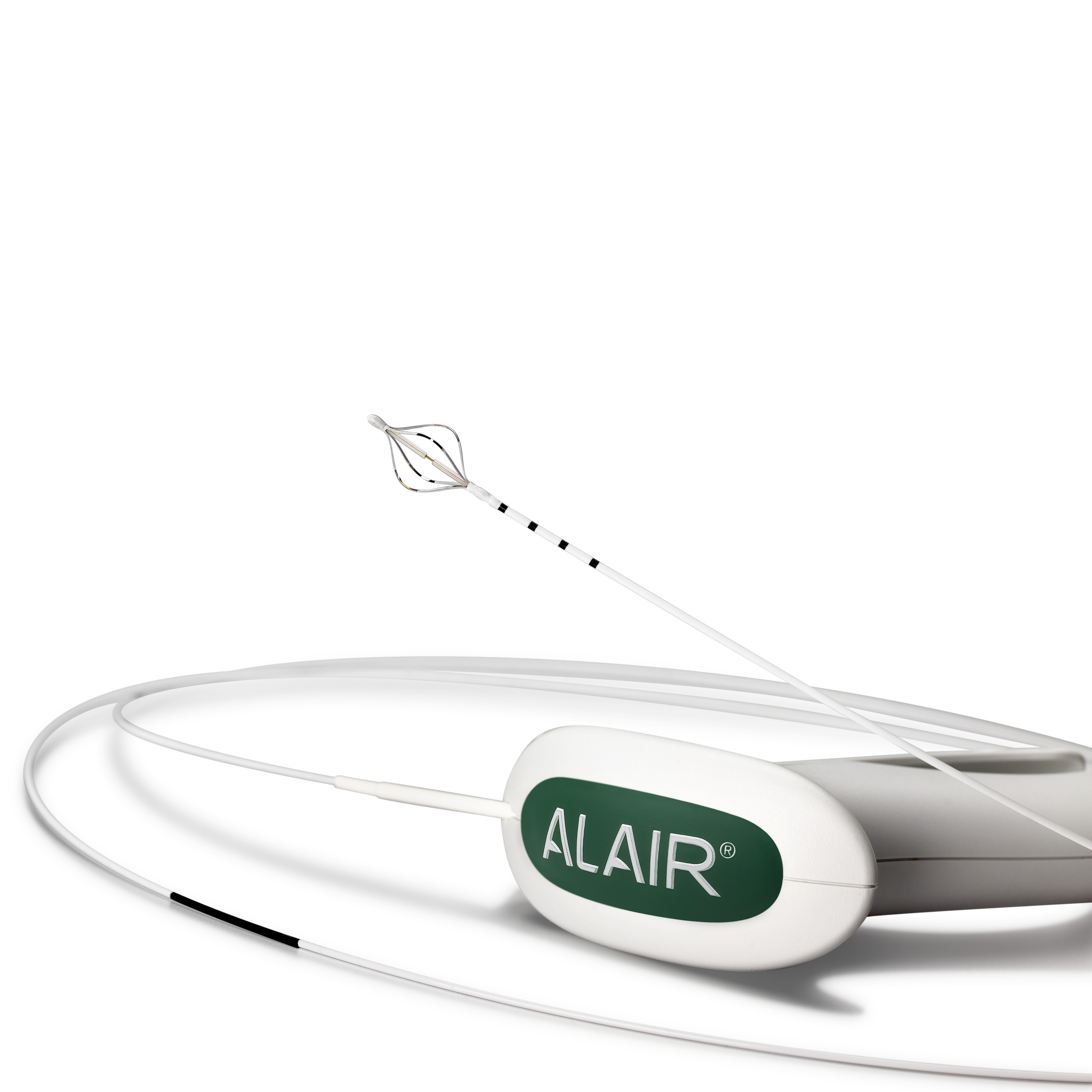
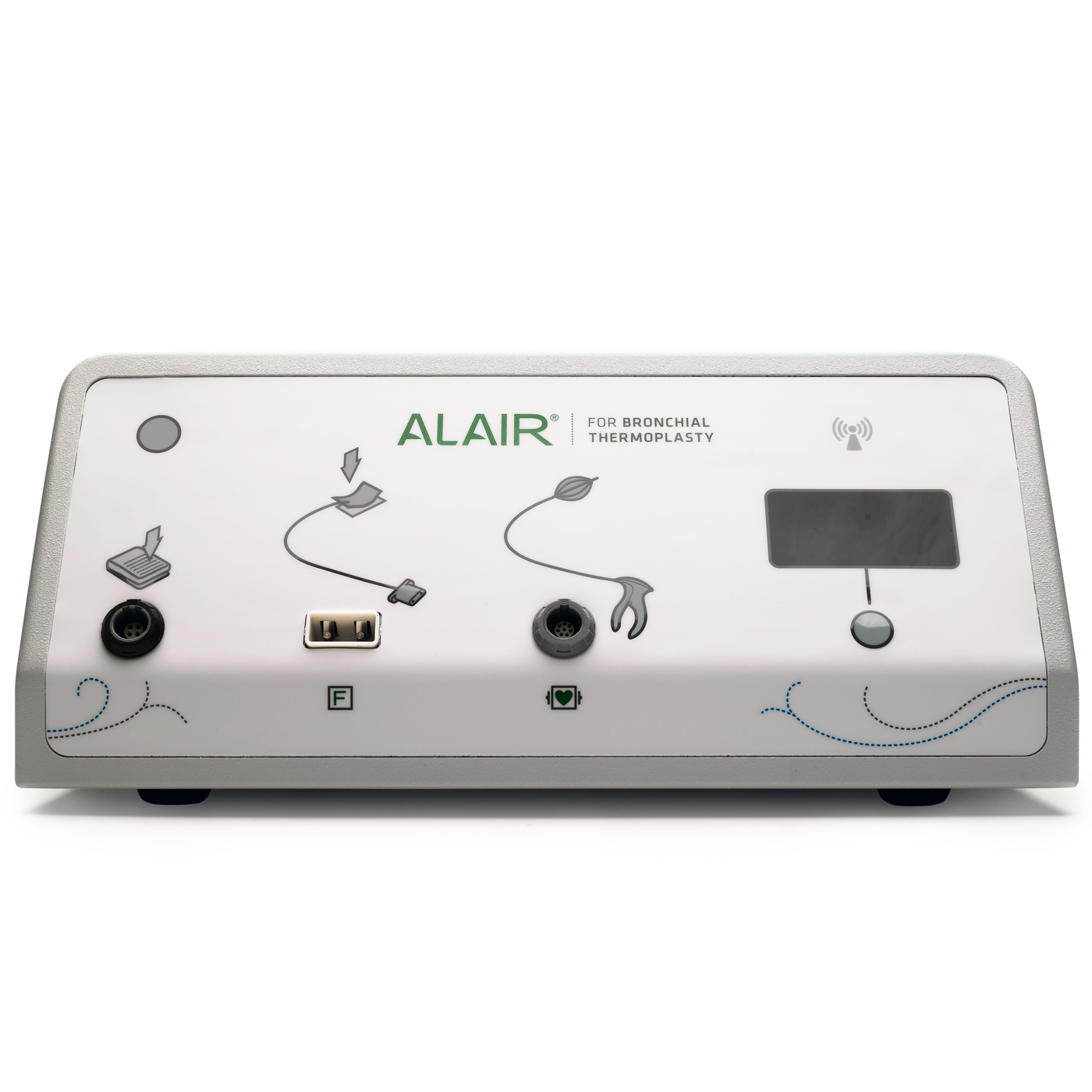
The Alair™ Bronchial Thermoplasty System
Bronchial Thermoplasty (BT), delivered by the Alair™ System, is a safe outpatient procedure for adult patients with severe asthma.
Explore
Product Details
Indications
Ordering Information
Alair™ Bronchial Thermoplasty System* |
|||
|---|---|---|---|
| Order Number | Model Number | Description | Packaging |
| M005ATS20000 | Alair ATS 200 | Alair Bronchial Thermoplasty Controller | Each |
| M005ATS25010 | Alair ATS 2-5 | Alair Bronchial Thermoplasty Catheter** | Each |
| M005ATS20110 | Alair ATS 201 | Alair RF Controller Accessory Kit | Each |
*Service Agreement available with 1, 2, 3, and 4 year terms. **Note: Initial stocking order requires a minimum order of 6 catheters (covering the complete treatment of 2 patients). |
|||
Physician & Patient Resources
Discover ACCESS
Read about the latest hot topics, news, and innovations in endoscopy. ACCESS is the online magazine for advancing the practice of endoscopy.
Find Education and Training
Helping you deliver the best patient care possible. EDUCARE makes it simple. One source. One stop. One schedule - yours.