References
1. PHYSICIAN'S TECHNICAL MANUAL RESONATE™ HF CRT-D, RESONATE™ CRT-D, RESONATE™ X4 CRT-D, VIGILANT™ CRT-D, VIGILANT™ X4 CRT-D, MOMENTUM™ CRT-D, MOMENTUM™ X4 CRT-D. Part Number: 51113879-001 EN US 2021-10-27
2. PHYSICIAN'S TECHNICAL MANUAL VISIONIST™, VISIONIST™ X4, VALITUDE™, VALITUDE™ X4, INTUA™, INVIVE™, CARDIAC RESYNCHRONIZATION THERAPY PACEMAKER. Part Number: 359252-003 EN US 2018-03-01
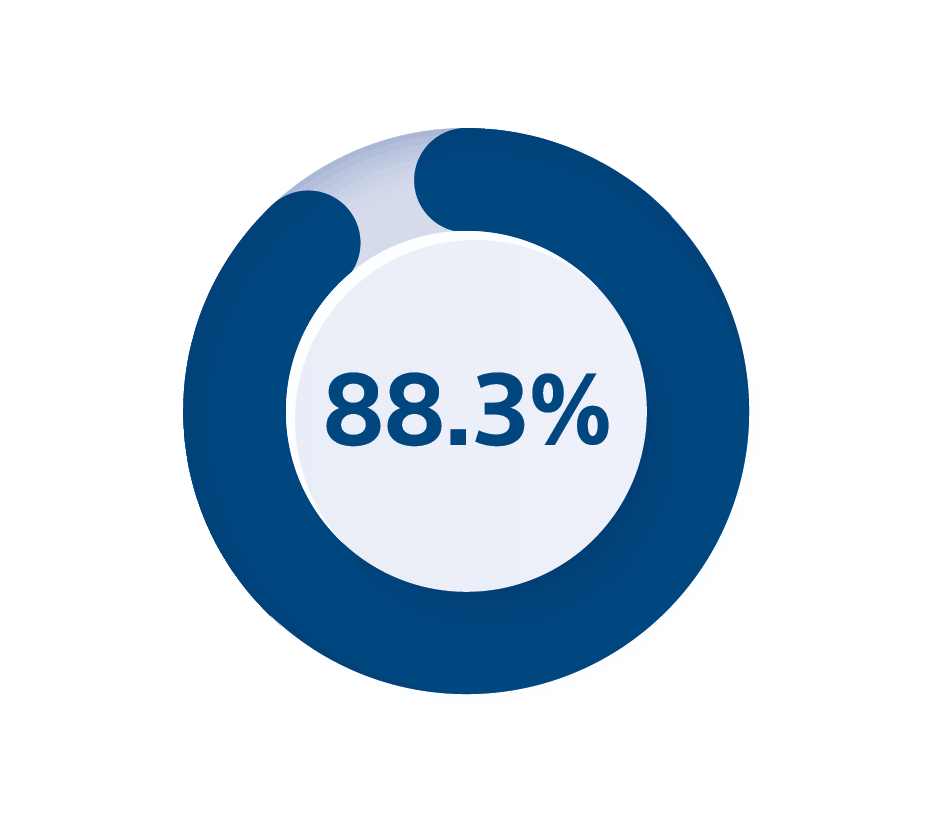
3. Boston Scientific Q4 2023 Product Performance Report survival probability including depletions and malfunctions of DYNAGEN/INOGEN/ORIGEN CRT-D at 8 years approved in the US April 2014, page 14, available online at HYPERLINK "http://www.bostonscientific.com/ppr"www.BostonScientific.com/ppr.
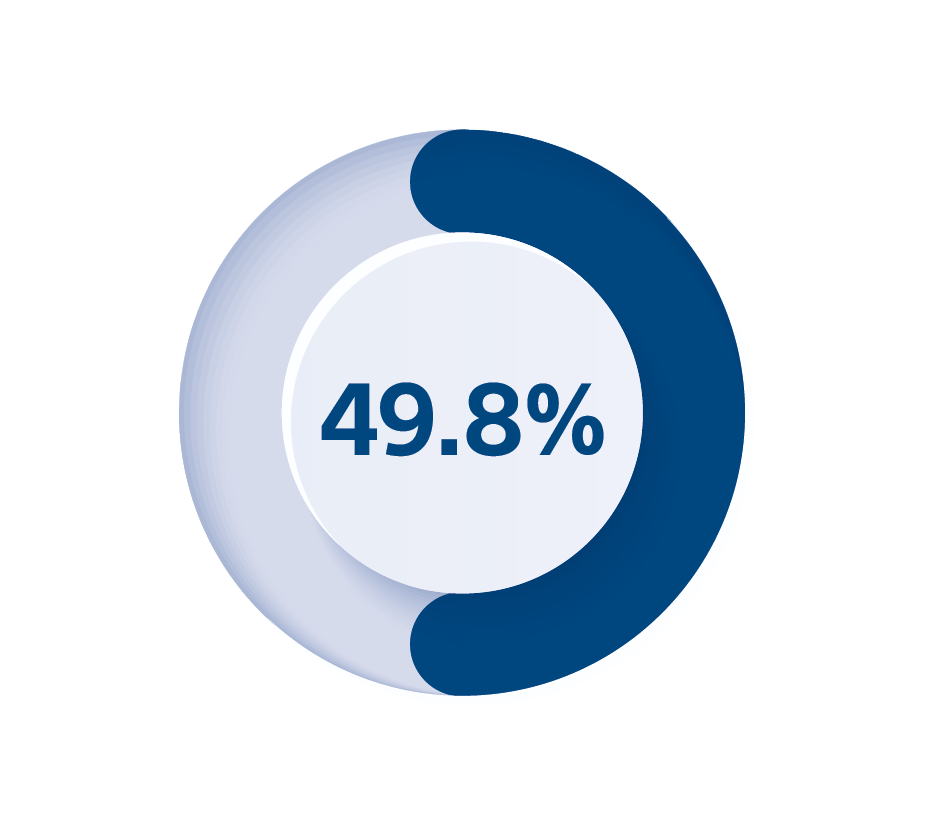
4. Medtronic CRM Product Performance Report survival probability including normal battery depletion of, DTBA1BD1 Viva XT CRT-D at 8 years and market released on 29 January 2013, Medtronic product performance as of 1 December 2023, available online at https://wwwp.medtronic.com/productperformance/. Viva is a trademark of Medtronic and its affiliates.
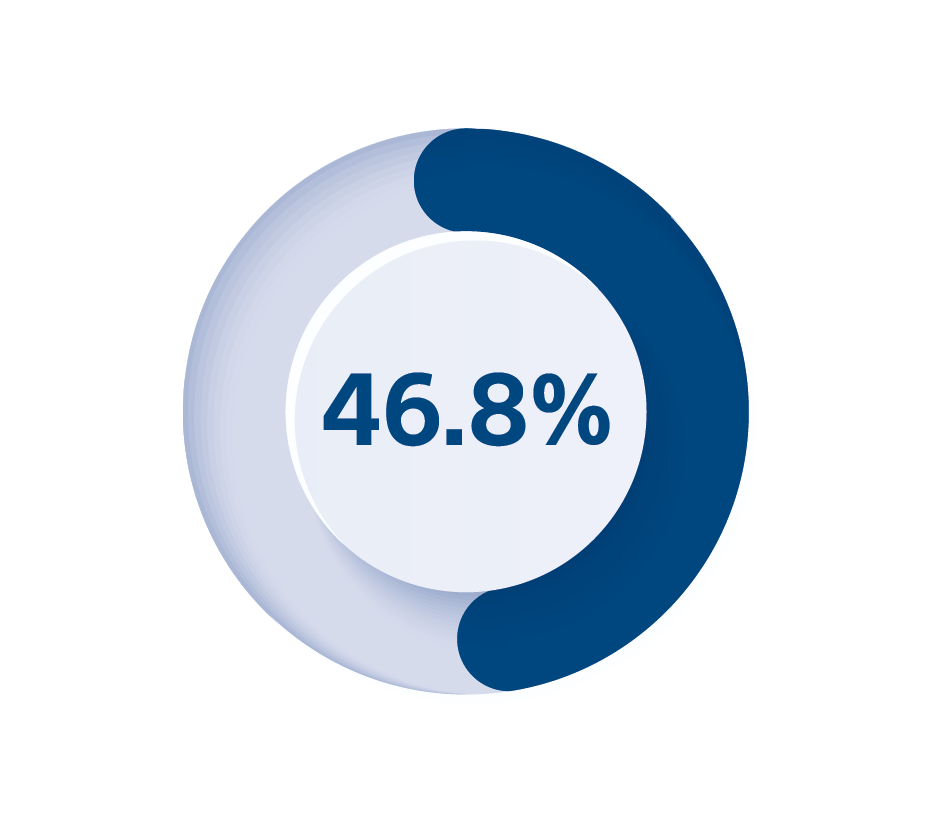
5. Medtronic CRM Product Performance Report survival probability including normal battery depletion of, DTBA1QQ Viva Quad XT CRT-D at 8 years and market released on 3 July 2014, Medtronic product performance as of 1 December 2023, available online at https://wwwp.medtronic.com/productperformance/. Viva is a trademark of Medtronic and its affiliates.