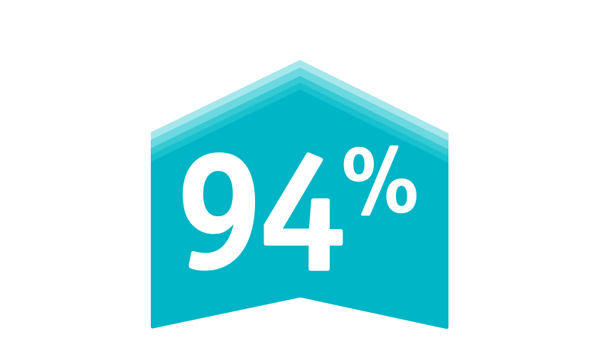
The PROTECTED TAVR Study is the largest randomized TAVR trial to date with 3,000 patients enrolled at >50 global sites who were randomized 1:1 – patients protected with SENTINEL vs. no use of SENTINEL during TAVR. All risk categories were eligible for inclusion, including low risk patients and all commercially available valves included as part of the trial.
Primary Endpoint: All Stroke (hemorrhagic, ischemic, or undetermined status; disabling or non-disabling) through 72-hours post TAVR procedure or hospital discharge.
Transient ischemic attack (TIA) and delirium were also reported on as part of the secondary neurological endpoints.
All patients enrolled in the trial underwent neurological examination at baseline and post TAVR procedure (discharge or 72-hours, whichever came first). This assessment was performed by a neurology professional (board certified/board eligible neurologist, neurology fellow, neurology physician assistant, or neurology nurse practitioner).