Pacemakers
Introducing ACCOLADE™ MRI and ESSENTIO™ MRI
Boston Scientific's most advanced pacemakers are now MR-conditional.
All Specialties (-)
-
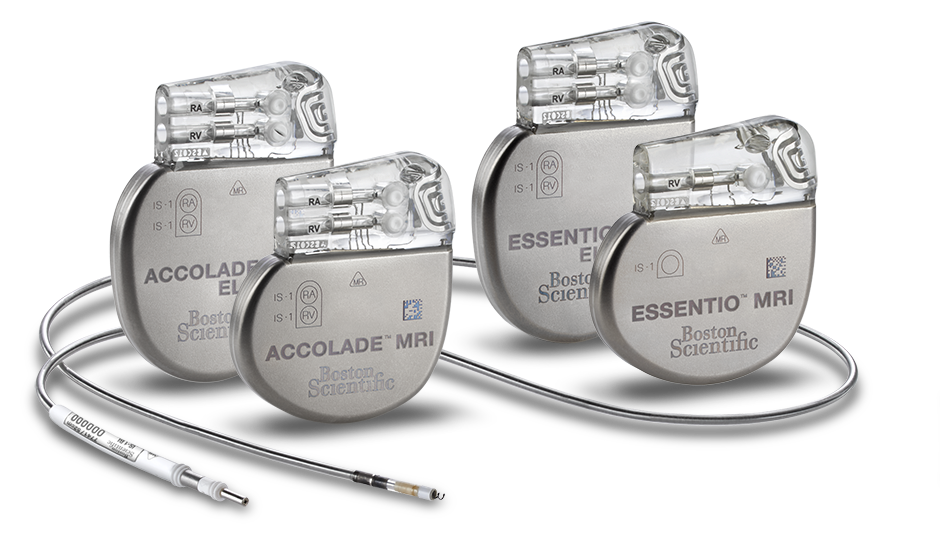
ACCOLADE™ and ESSENTIO™ Pacemakers
ACCOLADE and ESSENTIO offer RightRateTM Respiration-Based Pacing and Automatic Daily Monitoring to improve patient compliance.
- Electrophysiology
-
ACCOLADE™ MRI and ESSENTIO™ MRI Pacemakers
Boston Scientific's most advanced pacemakers are now MR-conditional. ACCOLADE MRI and ESSENTIO MRI models can be used as part of the ImageReady™ MR-Conditional Pacing System for safe and effective scanning in 1.5T and 3T MRI environments when MRI Conditions of Use are met.1
- Electrophysiology
-
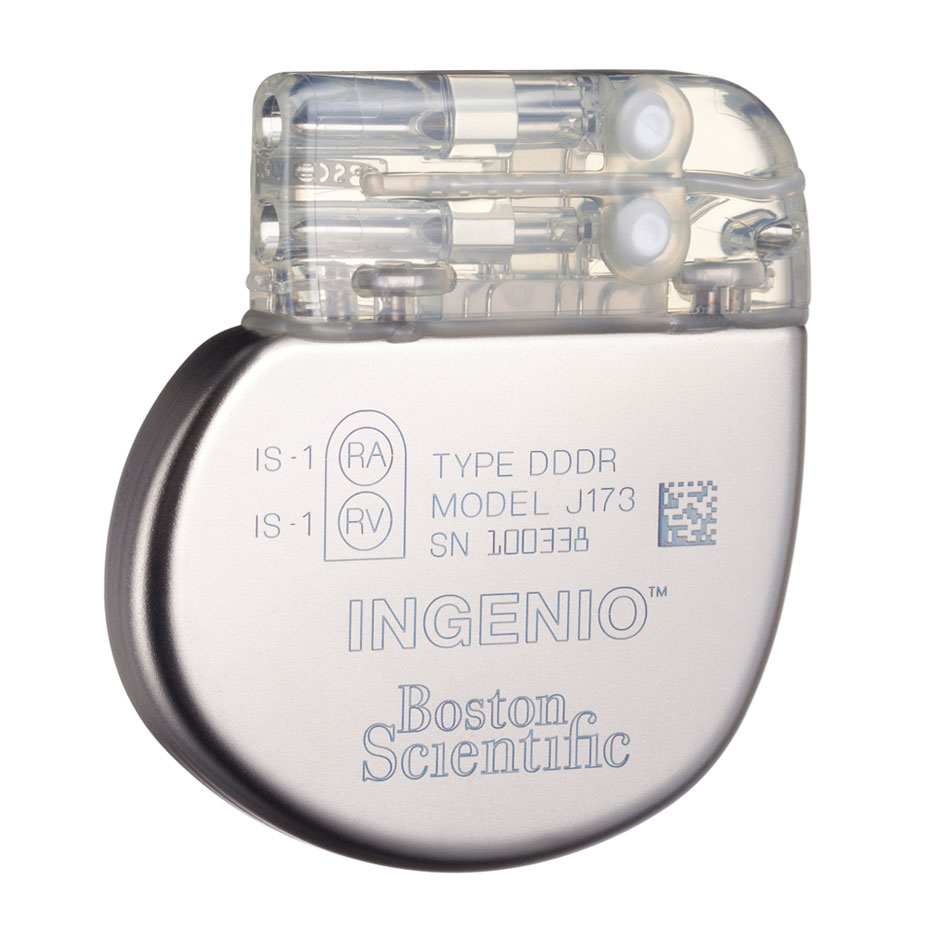
INGENIO™ and ADVANTIO™ Pacemakers
The INGENIO and ADVANTIO pacemakers from Boston Scientific define a new era in pacing. Both leverage key aspects of our innovative high-voltage platform and incorporate new features, therapies and diagnostic options.
- Electrophysiology
-
VITALIO™ Pacemaker
VITALIO lets the heart take the lead, intervening only when appropriate. With features including respiration-based pacing for rate response, wireless connectivity and automaticity in both chambers – the VITALIO pacemaker lets your patients get back to what’s important in life.
- Electrophysiology