EXALTTM Model D Single-Use Duodenoscope / Economic Impact
A cost-effective alternative to reusable duodenoscopes
The clinical data of the EXALT Model D Single-Use Duodenoscope is clear – and so is the economic impact on your facility and your practice. Model D can be an economically viable, cost-effective solution due to:
- Elimination of reprocessing costs associated with reusable duodenoscopes*
- Reduction of operational costs due to ineffective reprocessing and decreased risk of patient to patient contamination
*Assumes full conversion to EXALT Model D single-use duodenoscopes.

Avoid the hidden costs of reprocessing
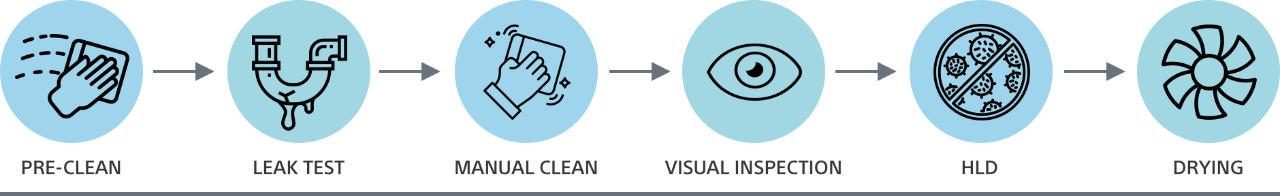
Properly reprocessing a reusable duodenoscope includes more than 100 distinct steps.1,2 From pre-cleaning to high-level disinfection to drying, each step requiring time and adding to costs. With EXALT Model D Single-Use Duodenoscope, you can drive down operational costs by reducing infection risk due to ineffective reprocessing and enabling staff to focus on other tasks.
Physician perspectives
Duodenoscope costs
Dr. Kenneth Binmoeller discusses the real-world costs and expenses of reusable versus single-use duodenoscopes.
![]()
Stay up to date
Sign up to receive periodic emails about EXALT Model D case studies, clinical data, reimbursement and more.
![]()
Connect with a rep
Request a rep to learn how EXALT Model D may help you address infection risks and improve patients’ lives.

