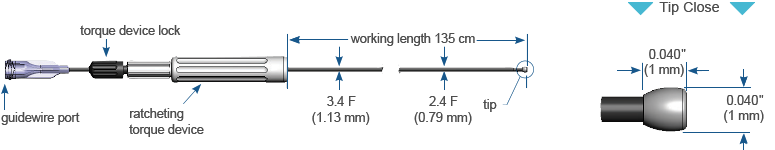
CrossBoss™
Coronary Catheter
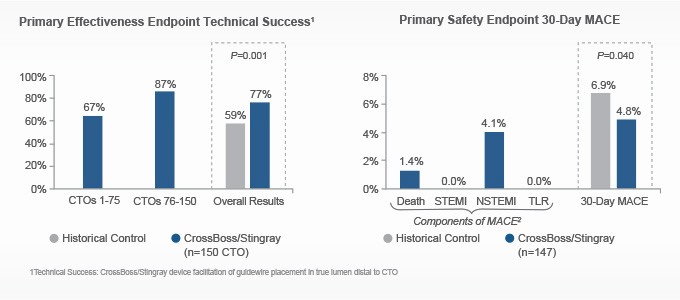
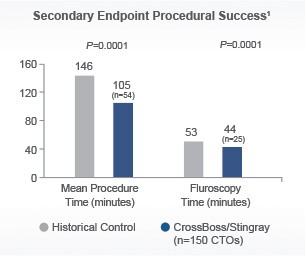
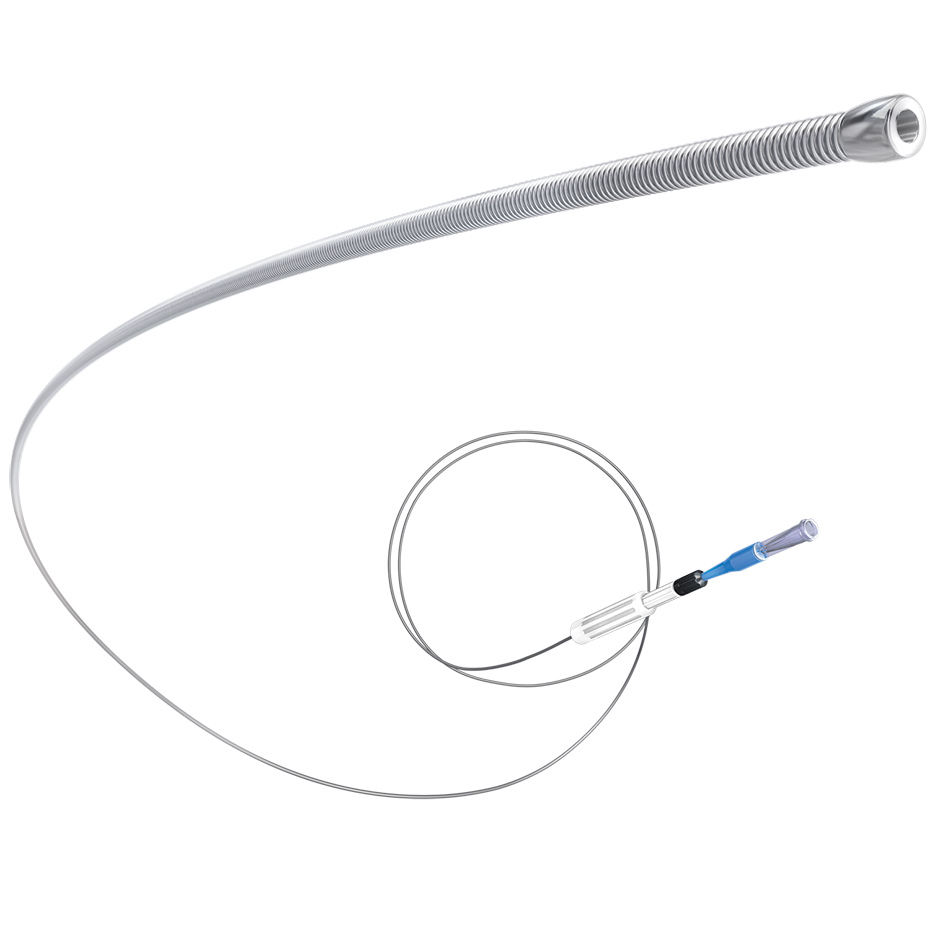
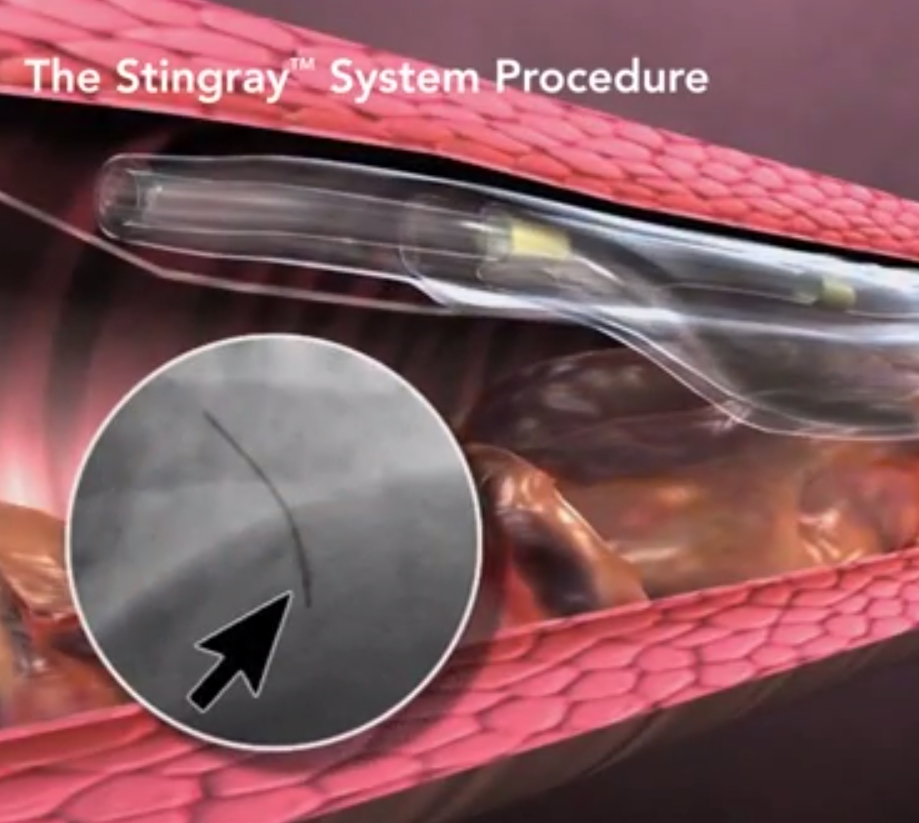
Designed to quickly and safely deliver a guidewire via the true lumen or subintimal pathways, CrossBoss Catheter gives you total access to coronary chronic total occlusions.
Learn more
Explore
Product Specifications
- Designed to quickly and safely deliver a guidewire via true lumen or subintimal pathways, CrossBoss gives you access to coronary chronic total occlusions.
- Atraumatic, rounded tip reduces risk of perforation
- Hydrophilic coated, multi-wire coiled shaft provides precise turn-for-turn response
- Fast-Spin torque device allows rapid rotation of the catheter to facilitate crossing
Ordering Information
| Product | ORDER CODE |
|---|---|
| CrossBoss Coronary Crossing Catheter | H749M2000B0 |
| StingRay Coronary Re-Entry System | Training required |
| StingRay Guidewire, 300cm | Training required |
| StingRay Guidewire, 185cm | Training required |
| StringRay Guidewire Extension | Training required |