AGENT™
Drug-Coated Balloon
Leave The Right AGENT Behind
The AGENT™ Drug-Coated Balloon (DCB) makes reliable drug transfer more deliverable. AGENT is unlike other DCBs on the market. It builds on the proven balloon innovation and powerful efficient drug eluting technology by Boston Scientific that you know and trust.
Key Resources
Explore
Reach complex lesions with ease
Make reliable drug transfer more deliverable
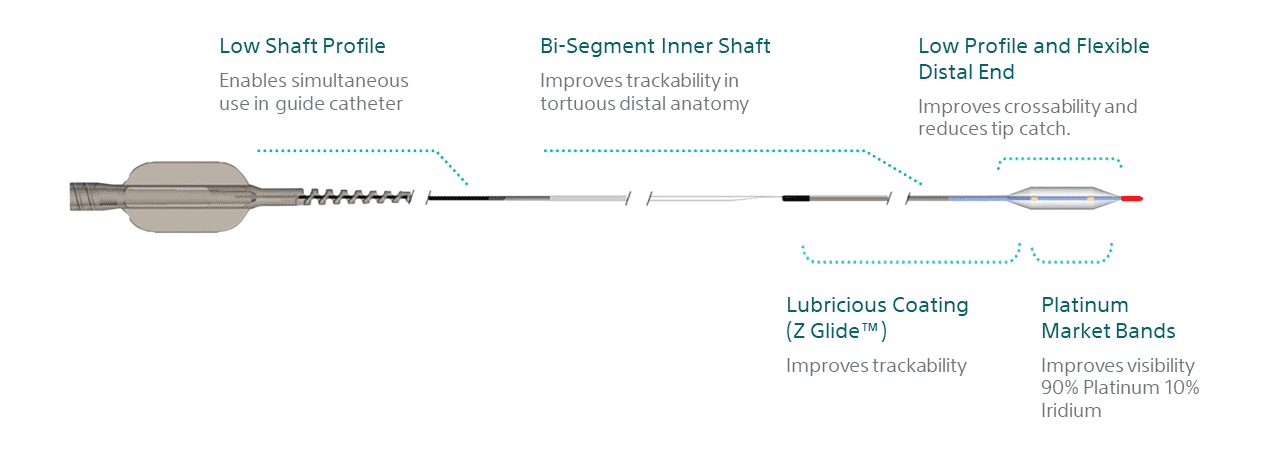
AGENT leverages the trusted EMERGE catheter for proven deliverability performance. Its bi-segment inner shaft improves flexibility without compromising push to ease delivery to distal lesions1. Furthermore, its ultra low tip profile is designed for crossability and reduced tip catch1.
Deliver the optimal dose when it matters most
AGENT has the most efficient drug transfer technology in the market with the lowest balloon drug dose and best-in-class acute transfer compared to other Drug Eluting Balloons (DEBs)2,3.
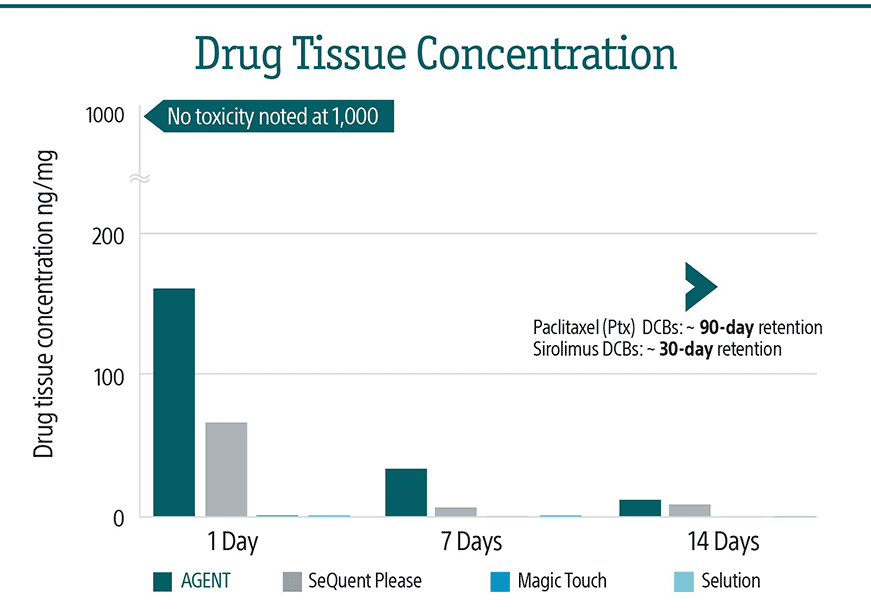
Three design features function together to provide reliable drug transfer.
Product Details
Product specifications
| Drug | Paclitaxel |
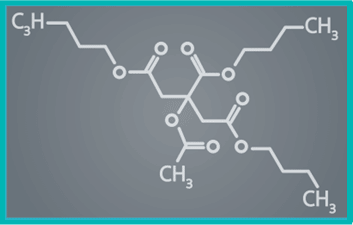
| Excipient | ATBC (Acetyl Tributyl Citrate) |
| Drug Dose | 2 μg/㎟ |
| Balloon Platform | EmergeTM |
| Entry Profile | ※0.0167 inch |
| Crossing Profile | ※0.91-1.14mm |
| Delivery System Effective Length | 144cm |
| Exchange Length | 26cm |
| Delivery System Port | Designated for 0.014" (0.36mm) Guidewire |
| Nominal Pressure | 6 atm |
| R.B.P. | 14 atm (2.0 – 3.25mm) |
| 12 atm (3.5mm/4.0mm) | |
| Guide Catheter Compatibility | 5F |
| Balloon Folds | 3 Wings |
| Catheter Shaft O.D. | Proximal 2.6F (≤0.91mm) Distal 2.3F (0.78mm)/2.4F (0.82mm) |
Compliance Chart
| ATM(kPa) | 2.00 | 2.25 | 2.50 | 2.75 | 3.00 | 3.50 | 4.00 |
| 3.0 (304) | 1.86 | 2.06 | 2.28 | 2.53 | 2.76 | 3.19 | 3.66 |
| 4.0 (405) | 1.93 | 2.14 | 2.37 | 2.61 | 2.85 | 3.30 | 3.80 |
| 5.0 (507) | 1.99 | 2.20 | 2.44 | 2.68 | 2.93 | 3.39 | 3.88 |
| 6.0 (608) | 2.03 | 2.26 | 2.50 | 2.75 | 3.00 | 3.46 | 3.96 |
| 7.0 (709) | 2.07 | 2.31 | 2.55 | 2.81 | 3.06 | 3.52 | 4.04 |
| 8.0 (811) | 2.10 | 2.34 | 2.59 | 2.85 | 3.11 | 3.57 | 4.09 |
| 9.0 (912) | 2.13 | 2.38 | 2.62 | 2.88 | 3.15 | 3.61 | 4.14 |
| 10.0 (1013) | 2.15 | 2.40 | 2.65 | 2.91 | 3.18 | 3.64 | 4.18 |
| 11.0 (1115) | 2.18 | 2.42 | 2.67 | 2.94 | 3.21 | 3.68 | 4.22 |
| 12.0 (1216) | 2.19 | 2.44 | 2.69 | 2.96 | 3.23 | 3.72 | 4.25 |
| 13.0 (1317) | 2.21 | 2.46 | 2.72 | 2.99 | 3.26 | | |
| 14.0 (1419) | 2.23 | 2.48 | 2.74 | 3.02 | 3.28 | | |
Ordering information
Balloon Length
| Ø(mm) | 12mm | 15mm | 20mm | 30mm |
|---|---|---|---|---|
| 2.00 | H74939222201210 | H74939222201510 | H74939222202010 | H74939222203010 |
| 2.25 | H74939222221210 | H74939222221510 | H74939222222010 | H74939222223010 |
| 2.50 | H74939222251210 | H74939222251510 | H74939222252010 | H74939222253010 |
| 2.75 | H74939222271210 | H74939222271510 | H74939222272010 | H74939222273010 |
| 3.00 | H74939222301210 | H74939222301510 | H74939222302010 | H74939222303010 |
| 3.50 | H74939222351210 | H74939222351510 | H74939222352010 | H74939222353010 |
| 4.00 | H74939222401210 | H74939222401510 | H74939222402010 | H74939222403010 |