InTouch Newsletter Issue 2

Congresses and Events
EuroPCR 2016: Presentation of COMET™ pressure wire by Prof. Escaned
COMET Pressure Guidewire provides streamlined workflow to facilitate decreased procedure duration so you can make the right decision the first time. Watch how Prof. Escaned simplified the treatment of Complex Multivessel disease with FFR guidance.
Building the next chapter in Structural Heart - Imaging and Intervention

In this webcast from EuroPCR 2016, a panel of experts explain the benefits of advanced imaging techniques in complex structural heart interventions. Watch the video and learn how paravalvular leak can be minimised despite anatomical complexities and how to expand the usage of LAA closure.
EuroPCR 2016: Stroke Prophylaxis in AF - When should you consider LAA Closure?

How do we understand the balance between periprocedural risk in the use of LAA closure as a prophylactic procedure? Join A. Vahanian, D. Foley and M.W.Bergmann to review our current evidence and clinical knowledge concerning LAA closure and stroke.
Educational Corner
European Expert Consensus on Rotational Atherectomy and Infographic

A group of highly experienced Rotablator operators released the first consensus statement establishing a basis for a standardised protocol for rotational atherectomy (RA). They highlight a selection of best practices, based on expert agreement of Rotablator use.
Learning with Clinical Cases:
Rotablator™ in Fibro-Calcified Lesions
Learning with Clinical Cases:
Tortuous Multivessel Disease Case with COMET™ (Dr Hill)
Clinical Studies
SYNERGY™ Stent: Addressing the Full Spectrum of Cardiovascular Disease Complexity
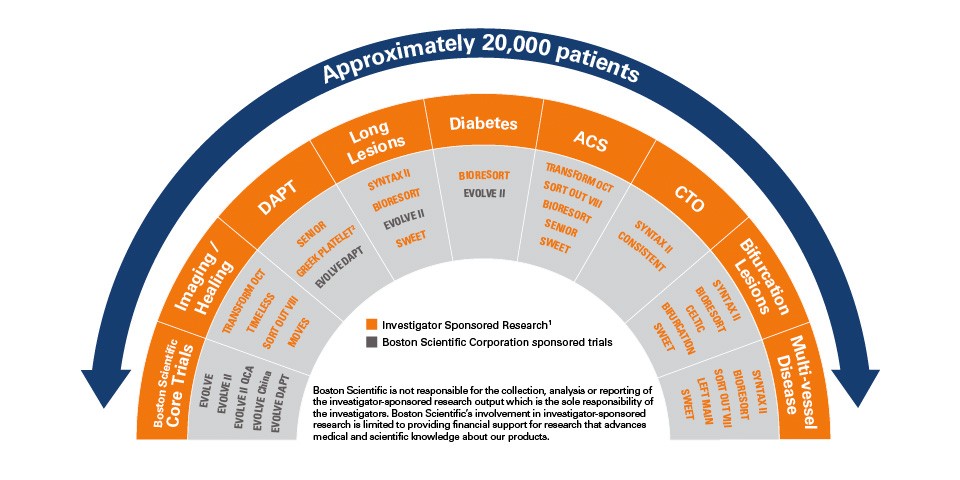
The SYNERGY™ Stent Clinical Program and research is studying approximately 20,000 patients in a wide variety of trials and will explore many types of patient populations. This research is designed to truly reflect the situations that interventional cardiologists experience every day in the cath lab.
Physician Experience

Global Chief-Medical-Officer, Dr. Keith Dawkins and U.S. Chief-Medical-Officer, Dr. Craig Thompson, discuss the compelling body of clinical and real-world evidence to support the safety of the SYNERGY™ Stent.
Safety Highlighted with the SYNERGY™ Stent System in the EVOLVE 5-Year Trial Data
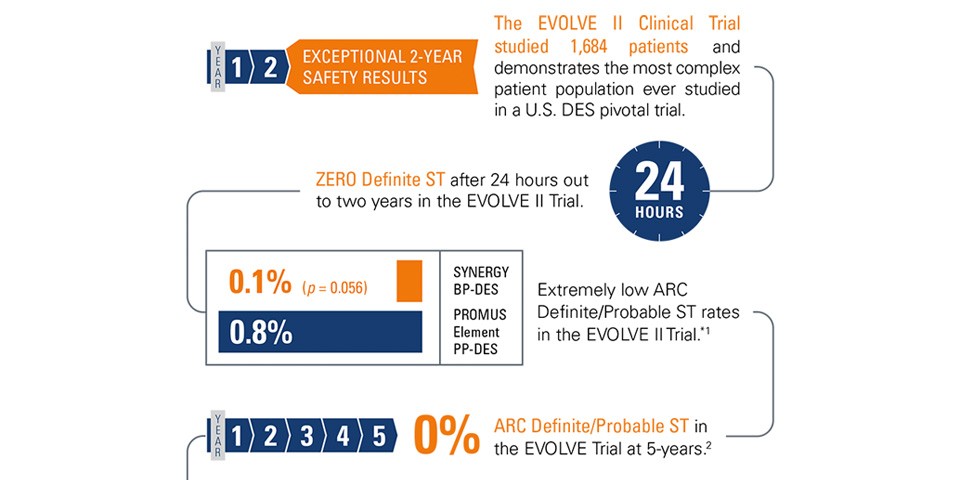
The EVOLVE 5-Year Data presented at EuroPCR 2016 showed the Long-term Safety with the SYNERGY™ Stent System.
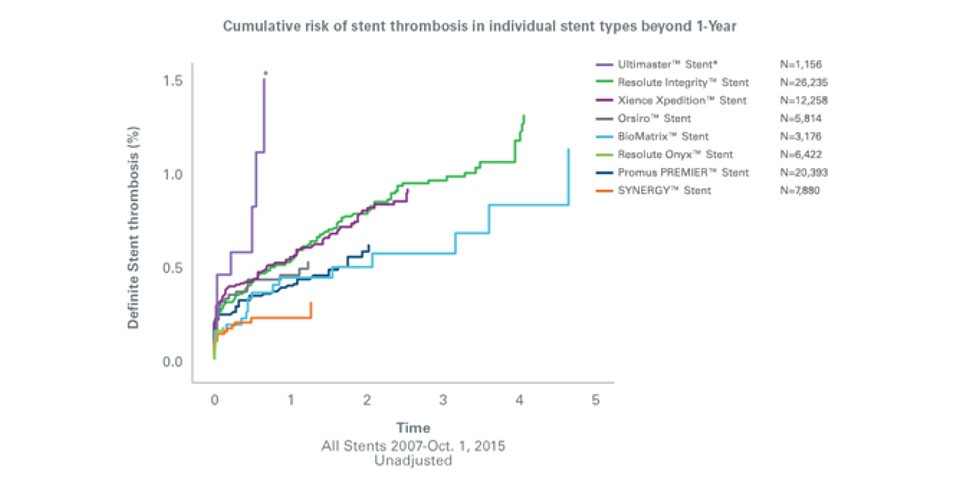
SYNERGY™ Stent reported lowest rates of ST in real-world SCAAR registry
In this real-world registry1 of over 83,000 patients, the SYNERGY™ stent’s thin strut design and bioabsorbable polymer coating show a trend of lower definite stent thrombosis compared to the other bioabsorbable polymer and permanent polymer drug-eluting stents.
SYNERGY™ Low Event Rates after Early DAPT Discontinuation in Complex Patients
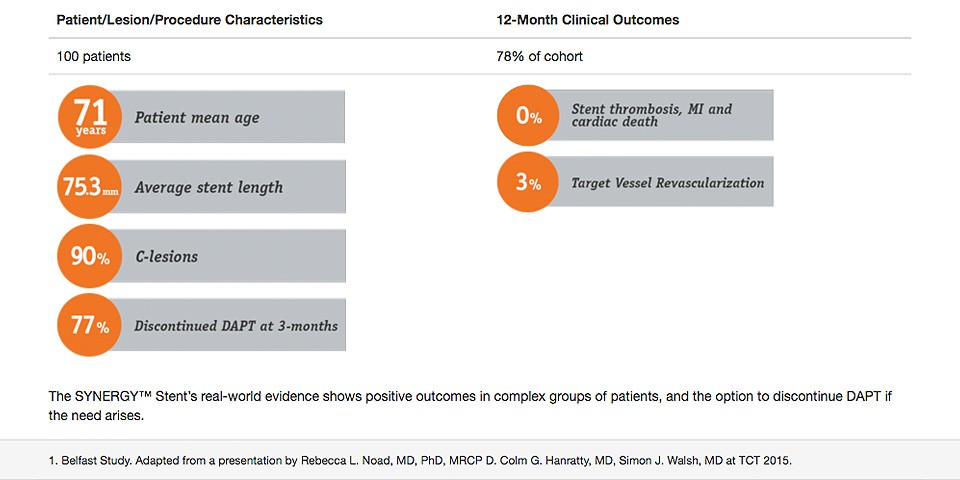
In a retrospective, single-center analysis of 100 extremely complex patients from Belfast, Ireland, the use of the SYNERGY™ stent allowed for early discontinuation of DAPT, without an increase in the incidence of stent thrombosis, MI and cardiac death at 12 Months.
















