S-ICD System IDE Clinical Study
Prospective, Single-Arm Comparison to Objective Performance Criteria
Pre-market Investigational Device Exemption (IDE)
Background
The implantable cardioverter-defibrillator (ICD) has been shown to reduce total mortality up to 54% and arrhythmic mortality 50% to 70% in randomized, multi-center studies.1-4
Complications associated with the ICD procedure and system may diminish the mortality benefit.5-8
The subcutaneous ICD (S-ICD™) was developed as an alternative system. The S-ICD is a completely subcutaneous system that does not require leads in the heart, leaving the vasculature untouched.
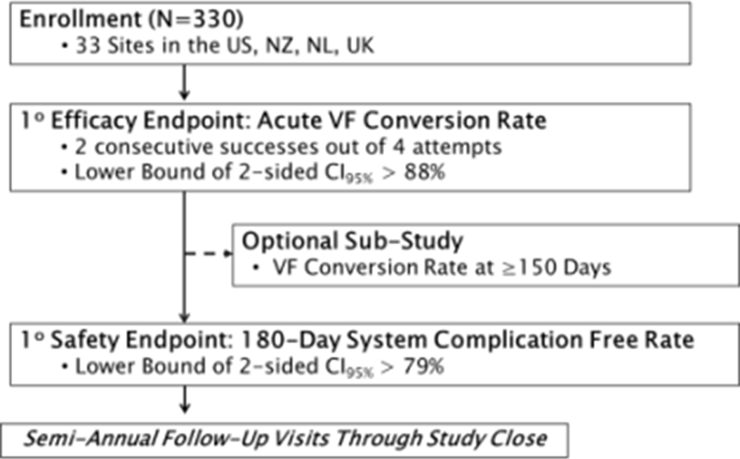
Study Design
Objective:
- Evaluate safety and effectiveness of the S-ICD System in the treatment of life-threatening ventricular arrhythmias
Design:
- Prospective, multicenter, single-arm clinical study conducted in the U.S., Europe, and New Zealand.
- Primary end-point comparison to objective performance criteria pre-defined by FDA
- Enrollment began Jan 2010 and concluded May 2011
- Follow up: > 1 year for first 100 patients; > 6 month for all patients
Inclusion
- Age ≥ 18 years
- Met guidelines for ICD implantation3 or replacement of an existing ICD system
- An appropriate pre-operative ECG
Key Exclusion Criteria
- Prior VT reliably terminated with anti-tachycardia pacing
- Existing epicardial patches or subcutaneous array
- Unipolar pacemakers
- Severe renal dysfunction (GFR ≤ 29)
Definition of Complications
- Type I: caused by the S-ICD System
- Type II: caused by the S-ICD System user’s manual or labeling of the S-ICD System
- Type III: not caused by the S-ICD System but would not have occurred in the absence of the implanted S-ICD System
Demographics
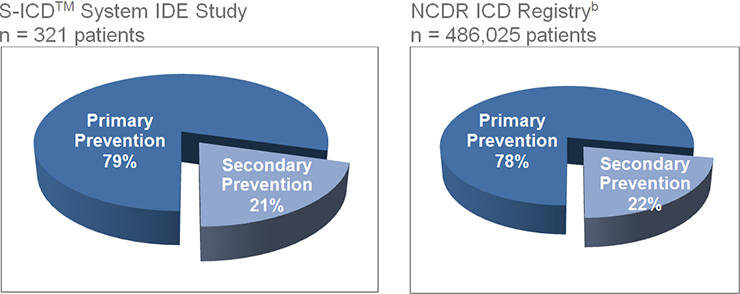
Attribute | Statistic/Category | N=321 | |
n | % | ||
| Co-morbidities History | Congestive Heart Failure | 197 | 61 |
Hypertension | 187 | 58 | |
Myocardial Infarction | 133 | 41 | |
Diabetes | 90 | 28 | |
Atrial Fibrillation | 49 | 15 | |
Valve Disease | 42 | 13 | |
COPD | 27 | 8 | |
Stroke | 18 | 6 | |
| Cardiac Surgical History | Percutaneous Revascularization | 92 | 29 |
CABG | 48 | 15 | |
TV-ICD | 43 | 13 | |
Valve Surgery | 18 | 6 | |
Pacemaker | 4 | 1 | |
Trial Results
Summary of key results
Effectiveness Endpoints Met
- 100% acute induced VF conversion rate
(75 patient sub-study tested at > 150 days) - 100% chronic induced VF conversion rate
- 95% of events treated in < 21 sec
- Mean time to therapy for all inductions was 14.6±2.9 seconds
Safety Endpoints Met
- 99% Free from Type I complications
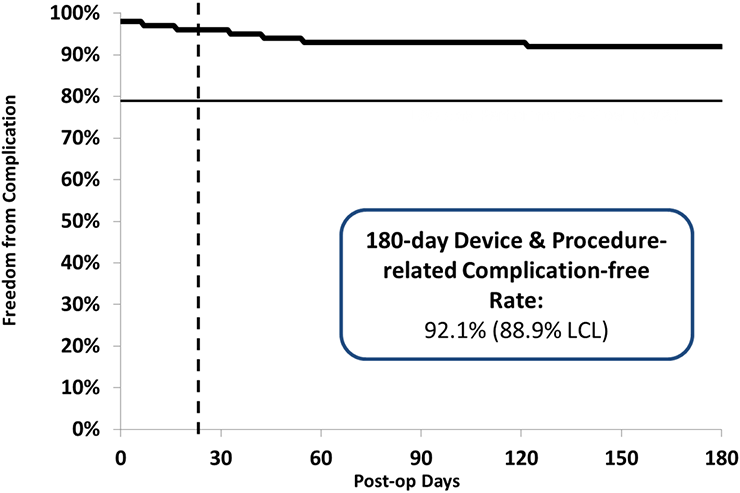
- 92.1% Free from Type I-III complications
- No deaths associated with S-ICD
Both endpoints met even under worst case sensitivity analysis
Induced VT/VF Conversion Results
| Evaluable Results | Conversion Rate (%) | 95% Lower Bound (%) | |
|---|---|---|---|
| Successful | Failure | ||
| 304 | 0 | 100.0 | 98.8 |
In 16 patients testing results were deemed nonevaluable and 1 patient did not undergo any testing because of persistent left ventricular thrombus. Ten of the 16 nonevaluable patients, including 1 patient not tested because of left ventricular thrombus, remained with the device and were followed up for the safety end point, whereas 7 patients were not implanted with the S-ICD System and were withdrawn from the study.
When all 17 excluded tests were classified as failures, the acute VF conversion rate had a success rate of 94.7% (lower 95% CI 91.7%) which exceeds the performance goal of 88%.
Spontaneous VF/VT Episodes
- 12 month first appropriate shock incidence = 6.2%
- 119 events in 21 patients
- 37 of 38 (97.4%) converted with with 1 or more shocks. The single episode not converted by the S-ICD System was an episode of monomorphic VT that terminated spontaneously while the device was charging to deliver a second shock.
- 92% first shock conversion efficacy
Spontaneous VT/VF Episodes | |||
|---|---|---|---|
Evaluation | Total | Discrete | Storm |
Episodes | 119 | 38 | 81 (4 events) |
Patients | 21* | 21 | 2 |
* Row entries do not sum because 2 pts experienced both discrete and storm episodes (mean follow up = 11 months)
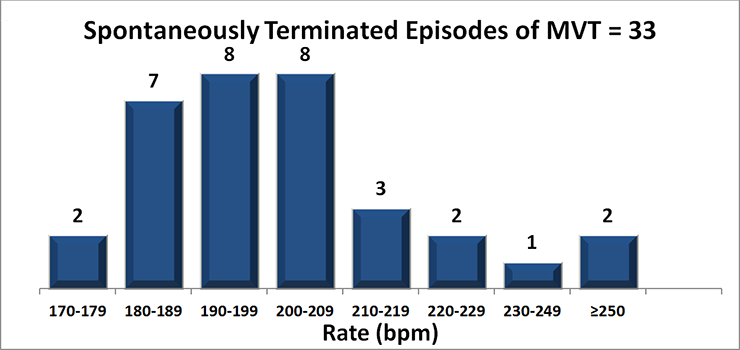
Therapy Appropriately Withheld
- Algorithm prevents therapy for VT/VF rhythms that spontaneously terminate prior to discharge.
- Therapy was appropriately withheld in 25 patients with 32 MVT episodes that met criteria to charge.
- No reports of syncope
Inappropriate Therapy
- 41 patients experienced shocks due to non-VT/VF events
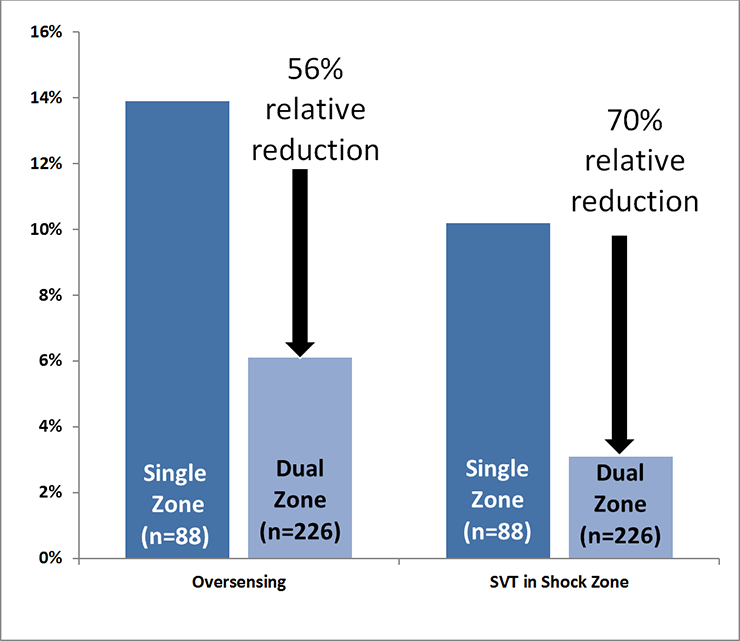
- Dual zone programming reduced inappropriate shocks
- No patients experienced a shock due to discrimination error in the Conditional Shock (dual) zone
Complications
- 4.4% perioperative complication rate
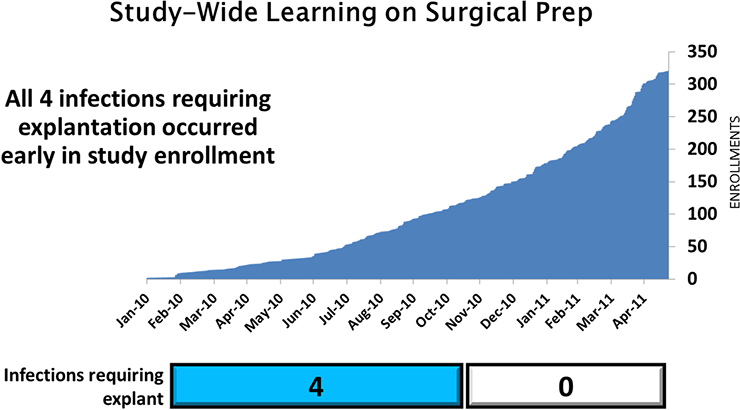
- 4 explant for infection (first 1/3 of pts)
- No arrhythmic deaths
- No endocarditis or systemic blood stream infections
Few Infections Led to Explant
- All infections requiring explant occurred early in the study before investigators meeting